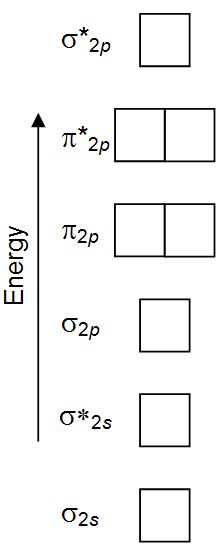
Using the arrows given below, complete the molecular orbital
occupancy for Ne2.
Do not leave any boxes unfilled.



Homework Answers

Add Answer to:
Using the arrows given below, complete the molecular orbital
occupancy for Ne2.
Do not leave any...
complete the molecular orbital for B2 with arrows given below and then predict its bond order....
 complete the molecular orbital for B2 with arrows given below
and then predict its bond order.
Complete the molecular orbital occupancy for B_2 with the arrows given below and then predict its bond order. Do not leave any boxes unfilled.
complete the molecular orbital for B2 with arrows given below
and then predict its bond order.
Complete the molecular orbital occupancy for B_2 with the arrows given below and then predict its bond order. Do not leave any boxes unfilled.
Draw molecular orbital energy level diagram for CH3 by following the steps given below. Valance orbital...
 Draw molecular orbital energy level diagram for CH3 by following the steps given below. Valance orbital ionization potentials: H(1s) - 13.6 eV, C(2s) -19.5 eV, C(2p) -10.7 ev. (6C, 11) E x +y?,z? 1 1 1 203 3C on 1 1 1 1 1 1 1 T - 1 0 2 1 1 -1 | 1 -1 1 -1 -1 TOT- 2 25; 1 1 - 1 1 -1 1 -1 1 30, 1 1 1 -1 0 1 -1...
Draw molecular orbital energy level diagram for CH3 by following the steps given below. Valance orbital ionization potentials: H(1s) - 13.6 eV, C(2s) -19.5 eV, C(2p) -10.7 ev. (6C, 11) E x +y?,z? 1 1 1 203 3C on 1 1 1 1 1 1 1 T - 1 0 2 1 1 -1 | 1 -1 1 -1 -1 TOT- 2 25; 1 1 - 1 1 -1 1 -1 1 30, 1 1 1 -1 0 1 -1...
Complete the orbital correlation diagram shown below by using e- (use arrows with the appropriate spin),...
 Complete the orbital correlation diagram shown below by using e- (use arrows with the appropriate spin), abel all hydrid (sp, sp2, sp3), atomic (# s, #p), and molecular orbitals (sigma, sigma*, pi, pi*), and circle the LUMO.
Complete the orbital correlation diagram shown below by using e- (use arrows with the appropriate spin), abel all hydrid (sp, sp2, sp3), atomic (# s, #p), and molecular orbitals (sigma, sigma*, pi, pi*), and circle the LUMO.
Using the molecular orbital diagram below, which of the following are paramagnetic? op 2P Energy 0...
 Using the molecular orbital diagram below, which of the following are paramagnetic? op 2P Energy 0 O A. Li2 0 OB. N2 C. F2 OD.CO OE. O2 2- OF. CN- OG. All of the above OH. None of the above
Using the molecular orbital diagram below, which of the following are paramagnetic? op 2P Energy 0 O A. Li2 0 OB. N2 C. F2 OD.CO OE. O2 2- OF. CN- OG. All of the above OH. None of the above
how do you find the values? i am confused Please complete the problems below related to...
 how do you find the values? i am confused
Please complete the problems below related to Bayes' Theorem. You must show any calculations where necessary to receive full credit Disease Present Disease Absent Test Positive 80 100 Test Negative 20 800 a. # of True Positives: b. # of True Negatives: c. # of False Positives: d, # of False Negatives: We were unable to transcribe this imageWe were unable to transcribe this image
how do you find the values? i am confused
Please complete the problems below related to Bayes' Theorem. You must show any calculations where necessary to receive full credit Disease Present Disease Absent Test Positive 80 100 Test Negative 20 800 a. # of True Positives: b. # of True Negatives: c. # of False Positives: d, # of False Negatives: We were unable to transcribe this imageWe were unable to transcribe this image
please answer question 7 using question 5 and the molecular orbital diagram given. 5. Prepare a...
 please answer
question 7 using question 5 and the molecular orbital diagram
given.
5. Prepare a molecular orbital diagram for BN. You may consider valence orbitals only. Label the atomic orbitals for B and N with the appropriate potential energy. Decide which orbitals have the correct symmetry, orientation, and potential energies to interact (see the handout provided during class). Label the valence atomic orbitals with the appropriate potential energy. Should s-p mixing be considered for BN? How will the MO...
please answer
question 7 using question 5 and the molecular orbital diagram
given.
5. Prepare a molecular orbital diagram for BN. You may consider valence orbitals only. Label the atomic orbitals for B and N with the appropriate potential energy. Decide which orbitals have the correct symmetry, orientation, and potential energies to interact (see the handout provided during class). Label the valence atomic orbitals with the appropriate potential energy. Should s-p mixing be considered for BN? How will the MO...
please help in all sections asap!! Which of the molecules below is the expected major product...
 please help in all sections asap!!
Which of the molecules below is the expected major product of the following reduction? H3C- CH3 Lindlar's catalyst HH Х НАС CH₃ HiC CH₂ We were unable to transcribe this imagePRINT Choose the correct reagents listed in the table below. 1)NBS, heat 2)NaOEt b AICI Na, CH3OH Hz, Lindlar's catalyst NH3 Conc. H2SO4 H2O+ 03. МСРВА DMS Enter the correct letter for each step of the reaction in the boxes below. (Reagents cannot be...
please help in all sections asap!!
Which of the molecules below is the expected major product of the following reduction? H3C- CH3 Lindlar's catalyst HH Х НАС CH₃ HiC CH₂ We were unable to transcribe this imagePRINT Choose the correct reagents listed in the table below. 1)NBS, heat 2)NaOEt b AICI Na, CH3OH Hz, Lindlar's catalyst NH3 Conc. H2SO4 H2O+ 03. МСРВА DMS Enter the correct letter for each step of the reaction in the boxes below. (Reagents cannot be...
Using a compass and straightedge only, do the following: a.) Construct the figure b.) Write out...
Using a compass and straightedge only, do the following: a.) Construct the figure b.) Write out step by step how to construct the figure 1.) Using a compass and straightedge only, construct a segment that is of length . Note, a segment of length 1 is given below and must be used. We were unable to transcribe this imageWe were unable to transcribe this image
please answer question 8 using question 5 and the molecular orbital diagram given. 5. Prepare a...
 please answer
question 8 using question 5 and the molecular orbital diagram
given.
5. Prepare a molecular orbital diagram for BN. You may consider valence orbitals only Label the atomic orbitals for B and N with the appropriate potential energy. Decide which orbitals have the correct symmetry, orientation, and potential energies to interact (see the handout provided during class). Label the valence atomic orbitals with the appropriate potential energy. Should s-p mixing be considered for BN? How will the MO...
please answer
question 8 using question 5 and the molecular orbital diagram
given.
5. Prepare a molecular orbital diagram for BN. You may consider valence orbitals only Label the atomic orbitals for B and N with the appropriate potential energy. Decide which orbitals have the correct symmetry, orientation, and potential energies to interact (see the handout provided during class). Label the valence atomic orbitals with the appropriate potential energy. Should s-p mixing be considered for BN? How will the MO...
Given the matrix fine all the values of a that make the = 0. Give your answers in increasing order. a can be _________ , 3.723 , or _________ note: leave any unneeded answer spaces blank. I know th...
Given the matrix fine all the values of a that make the = 0. Give your answers in increasing order. a can be _________ , 3.723 , or _________ note: leave any unneeded answer spaces blank. I know the value i have is right, but I can't seem to get the other number(s). -3.223 is not an answer. 59a 4 4 We were unable to transcribe this image 59a 4 4
Most questions answered within 3 hours.
-
Calculate the molality, mole-fraction and percent mass of 28.9M
HF at 25 degrees Celcius of the...
asked 3 minutes ago -
A developmental psychologist believes that children raised in
bilingual families will have higher verbal fluency at...
asked 10 minutes ago -
A fast food meal has 5660 kJ of energy. A person uses energy at
a rate...
asked 22 minutes ago -
The pKb for a generic amine(R-NH2)) in
aqueous solution is 6.30. What is its pKa?
asked 23 minutes ago -
The following reactions have the indicated equilibrium constants
at a particular temperature: N2(g) + O2(g) ⇌...
asked 26 minutes ago -
Please Help ASAP.
1Consider the below code which iterates over a linked
list of n nodes...
asked 42 minutes ago -
Determine the air to fuel ratio of:
Canadian natural gas, with 93.9% methane, 4.2% ethane, 0.3%...
asked 42 minutes ago -
A manufacturer of power tools claims that the average amount of
time required to assemble their...
asked 44 minutes ago -
"electron-withdrawing substituents on carbon encourage back
donation", then on the next page he says that "greater...
asked 56 minutes ago -
On December 31, 2016, the shareholders’ equity section of the
balance sheet of R & L...
asked 1 hour ago -
16.7
At t=0s a small "upward" (positive y) pulse centered at x = 5.0
m is...
asked 1 hour ago -
Twitter Users and News: A poll conducted in 2013 found that 52%
of U.S. adult Twitter...
asked 1 hour ago
 complete the molecular orbital for B2 with arrows given below
and then predict its bond order.
Complete the molecular orbital occupancy for B_2 with the arrows given below and then predict its bond order. Do not leave any boxes unfilled.
complete the molecular orbital for B2 with arrows given below
and then predict its bond order.
Complete the molecular orbital occupancy for B_2 with the arrows given below and then predict its bond order. Do not leave any boxes unfilled.
 Draw molecular orbital energy level diagram for CH3 by following the steps given below. Valance orbital ionization potentials: H(1s) - 13.6 eV, C(2s) -19.5 eV, C(2p) -10.7 ev. (6C, 11) E x +y?,z? 1 1 1 203 3C on 1 1 1 1 1 1 1 T - 1 0 2 1 1 -1 | 1 -1 1 -1 -1 TOT- 2 25; 1 1 - 1 1 -1 1 -1 1 30, 1 1 1 -1 0 1 -1...
Draw molecular orbital energy level diagram for CH3 by following the steps given below. Valance orbital ionization potentials: H(1s) - 13.6 eV, C(2s) -19.5 eV, C(2p) -10.7 ev. (6C, 11) E x +y?,z? 1 1 1 203 3C on 1 1 1 1 1 1 1 T - 1 0 2 1 1 -1 | 1 -1 1 -1 -1 TOT- 2 25; 1 1 - 1 1 -1 1 -1 1 30, 1 1 1 -1 0 1 -1...
 Complete the orbital correlation diagram shown below by using e- (use arrows with the appropriate spin), abel all hydrid (sp, sp2, sp3), atomic (# s, #p), and molecular orbitals (sigma, sigma*, pi, pi*), and circle the LUMO.
Complete the orbital correlation diagram shown below by using e- (use arrows with the appropriate spin), abel all hydrid (sp, sp2, sp3), atomic (# s, #p), and molecular orbitals (sigma, sigma*, pi, pi*), and circle the LUMO.
 Using the molecular orbital diagram below, which of the following are paramagnetic? op 2P Energy 0 O A. Li2 0 OB. N2 C. F2 OD.CO OE. O2 2- OF. CN- OG. All of the above OH. None of the above
Using the molecular orbital diagram below, which of the following are paramagnetic? op 2P Energy 0 O A. Li2 0 OB. N2 C. F2 OD.CO OE. O2 2- OF. CN- OG. All of the above OH. None of the above
 how do you find the values? i am confused
Please complete the problems below related to Bayes' Theorem. You must show any calculations where necessary to receive full credit Disease Present Disease Absent Test Positive 80 100 Test Negative 20 800 a. # of True Positives: b. # of True Negatives: c. # of False Positives: d, # of False Negatives: We were unable to transcribe this imageWe were unable to transcribe this image
how do you find the values? i am confused
Please complete the problems below related to Bayes' Theorem. You must show any calculations where necessary to receive full credit Disease Present Disease Absent Test Positive 80 100 Test Negative 20 800 a. # of True Positives: b. # of True Negatives: c. # of False Positives: d, # of False Negatives: We were unable to transcribe this imageWe were unable to transcribe this image
 please answer
question 7 using question 5 and the molecular orbital diagram
given.
5. Prepare a molecular orbital diagram for BN. You may consider valence orbitals only. Label the atomic orbitals for B and N with the appropriate potential energy. Decide which orbitals have the correct symmetry, orientation, and potential energies to interact (see the handout provided during class). Label the valence atomic orbitals with the appropriate potential energy. Should s-p mixing be considered for BN? How will the MO...
please answer
question 7 using question 5 and the molecular orbital diagram
given.
5. Prepare a molecular orbital diagram for BN. You may consider valence orbitals only. Label the atomic orbitals for B and N with the appropriate potential energy. Decide which orbitals have the correct symmetry, orientation, and potential energies to interact (see the handout provided during class). Label the valence atomic orbitals with the appropriate potential energy. Should s-p mixing be considered for BN? How will the MO...
 please help in all sections asap!!
Which of the molecules below is the expected major product of the following reduction? H3C- CH3 Lindlar's catalyst HH Х НАС CH₃ HiC CH₂ We were unable to transcribe this imagePRINT Choose the correct reagents listed in the table below. 1)NBS, heat 2)NaOEt b AICI Na, CH3OH Hz, Lindlar's catalyst NH3 Conc. H2SO4 H2O+ 03. МСРВА DMS Enter the correct letter for each step of the reaction in the boxes below. (Reagents cannot be...
please help in all sections asap!!
Which of the molecules below is the expected major product of the following reduction? H3C- CH3 Lindlar's catalyst HH Х НАС CH₃ HiC CH₂ We were unable to transcribe this imagePRINT Choose the correct reagents listed in the table below. 1)NBS, heat 2)NaOEt b AICI Na, CH3OH Hz, Lindlar's catalyst NH3 Conc. H2SO4 H2O+ 03. МСРВА DMS Enter the correct letter for each step of the reaction in the boxes below. (Reagents cannot be...
 please answer
question 8 using question 5 and the molecular orbital diagram
given.
5. Prepare a molecular orbital diagram for BN. You may consider valence orbitals only Label the atomic orbitals for B and N with the appropriate potential energy. Decide which orbitals have the correct symmetry, orientation, and potential energies to interact (see the handout provided during class). Label the valence atomic orbitals with the appropriate potential energy. Should s-p mixing be considered for BN? How will the MO...
please answer
question 8 using question 5 and the molecular orbital diagram
given.
5. Prepare a molecular orbital diagram for BN. You may consider valence orbitals only Label the atomic orbitals for B and N with the appropriate potential energy. Decide which orbitals have the correct symmetry, orientation, and potential energies to interact (see the handout provided during class). Label the valence atomic orbitals with the appropriate potential energy. Should s-p mixing be considered for BN? How will the MO...