Please draw it on paper instead of predicting the products in a chemical equation form.
Homework Answers
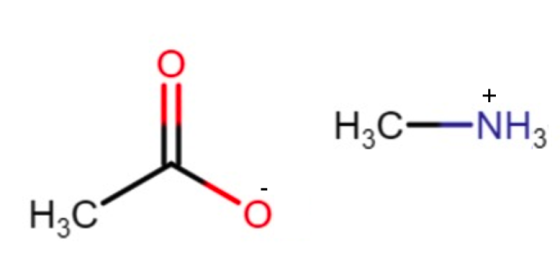
CH3COOH is an acid and CH3-CH2 is base
CH3COOH will loose H+ to form CH3COO-
CH3-NH2 will gain H+ to form CH3-NH3+
They are drawn below:
Add Answer to:
Please draw it on paper instead of predicting the products in a chemical equation form. Predict...
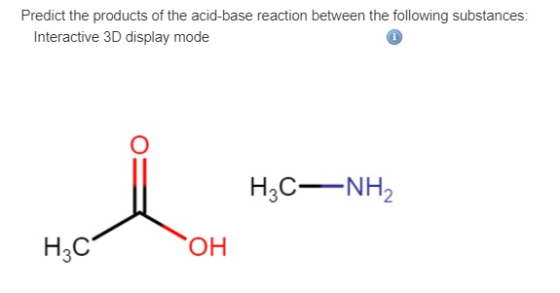
Predict the products of the acid-base reaction between the following substances: Interactive 3D display mode Draw...
 Predict the products of the acid-base reaction between the following substances: Interactive 3D display mode Draw the molecules or ions on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced Template toolbars, including charges where needed. The single bond is active by default.
Predict the products of the acid-base reaction between the following substances: Interactive 3D display mode Draw the molecules or ions on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced Template toolbars, including charges where needed. The single bond is active by default.
Draw the products of the acid-base reaction between the following species: Interactive 3D display mode Draw...
 Draw the products of the acid-base reaction between the following species: Interactive 3D display mode Draw the products on the canvas by choosing buttons from the Tools (for bonds), Atoms
Draw the products of the acid-base reaction between the following species: Interactive 3D display mode Draw the products on the canvas by choosing buttons from the Tools (for bonds), Atoms
Predict the products of the acid-base reaction between the following substances: *Draw* the molecules or ions....
 Predict the products of the acid-base reaction between the
following substances:
*Draw* the molecules or ions.
Predict the products of the acid-base reaction between the following substances: Draw the molecules or ions.
Predict the products of the acid-base reaction between the
following substances:
*Draw* the molecules or ions.
Predict the products of the acid-base reaction between the following substances: Draw the molecules or ions.
Part A Predict the products of the acid-base reaction between the following substances: Interactive MarvinView
 Part A Predict the products of the acid-base reaction between the following substances: Interactive MarvinView
Part A Predict the products of the acid-base reaction between the following substances: Interactive MarvinView
Draw the keto tautomeric form of the following compounds: Part A Draw the keto tautomeric form...
 Draw the keto tautomeric form of the following compounds:
Part A Draw the keto tautomeric form of the following compound Interactive 3D display mode H3C CH2 ÓH
Draw the keto tautomeric form of the following compounds:
Part A Draw the keto tautomeric form of the following compound Interactive 3D display mode H3C CH2 ÓH
Draw the organic product of the acid-base reaction between the following species: Interactive 3D display mode...
 Draw the organic product of the acid-base reaction between the following species: Interactive 3D display mode Draw the molecule or ion on the canvas by choosing buttons from the Tools (for bonds), Atoms and Advanced Template toolbars, including charges where needed. The single bond is active b default.
Draw the organic product of the acid-base reaction between the following species: Interactive 3D display mode Draw the molecule or ion on the canvas by choosing buttons from the Tools (for bonds), Atoms and Advanced Template toolbars, including charges where needed. The single bond is active b default.
Please Help! Draw the major product of the following reaction: Interactive 3D display mode HN CI
 Please Help!
Draw the major product of the following reaction: Interactive 3D display mode HN CI
Please Help!
Draw the major product of the following reaction: Interactive 3D display mode HN CI
yeAgtiCicHDafV2XDloDXmVc7271oBw7QYban Investments waigreens O SIMPLE REACTIONS Predicting the products of a neutralization reaction Predict the products...
 yeAgtiCicHDafV2XDloDXmVc7271oBw7QYban Investments waigreens O SIMPLE REACTIONS Predicting the products of a neutralization reaction Predict the products of the reaction below. That is, complete the right-hand side of the chemical equation. Be sure your equation is balanced. HBrCa(OH), ? X
yeAgtiCicHDafV2XDloDXmVc7271oBw7QYban Investments waigreens O SIMPLE REACTIONS Predicting the products of a neutralization reaction Predict the products of the reaction below. That is, complete the right-hand side of the chemical equation. Be sure your equation is balanced. HBrCa(OH), ? X
Predict the products that could form. Next to the products you draw, right either “products not...
 Predict the products that could form. Next to the products you
draw, right either “products not favored” or “products
favored”
4. This question continues on the next page. For each of the following acid-base reactions: (0.3 pts ea = 3.0 pts) a. Predict the products that could form. b. Next to the products you draw, write either "Products not favored" or "Products favored." f. HCECH + CH3Li →
Predict the products that could form. Next to the products you
draw, right either “products not favored” or “products
favored”
4. This question continues on the next page. For each of the following acid-base reactions: (0.3 pts ea = 3.0 pts) a. Predict the products that could form. b. Next to the products you draw, write either "Products not favored" or "Products favored." f. HCECH + CH3Li →
Draw all possible products formed when the compound shown below undergoes reaction with 1 equivalent of...
 Draw all possible products formed when the compound shown below
undergoes reaction with 1 equivalent of Br2.
Interactive 3D display mode
Draw all possible products formed when the compound shown below
undergoes reaction with 1 equivalent of Br2.
Interactive 3D display mode
Most questions answered within 3 hours.
-
Calculate and plot the number and weight distributions of x-mers
found in a step-growth polymerization for...
asked 1 minute ago -
The Baily Corporation has developed a specialized software
program that improves inventory control capability. The following...
asked 4 minutes ago -
Problem 5-4A (Part Level Submission) Wolford Department Store is
located in midtown Metropolis. During the past...
asked 4 minutes ago -
Preparation of Benzoic Acid using a Grignard Reagent URGENT
1. During your Grignard formation, a small...
asked 27 minutes ago -
A uniform magnetic field is perpendicular to the plane of a wire
loop. If the loop...
asked 26 minutes ago -
At the peak of your career, your were earning $120,000 and
holding a top level position....
asked 29 minutes ago -
. A permanent magnet is dropped south-end-down through a horizontal
circular coil with a radius of...
asked 32 minutes ago -
Bernie's Beverages purchased some fixed assets classified as
5-year property for MACRS. The assets cost $28,000....
asked 46 minutes ago -
How many ATPs are produced from the catabolism of a 10-C
molecule of fatty acid under...
asked 50 minutes ago -
Before practicing a routine on the rings, a 64.8 kg gymnast
hangs motionless, with one hand...
asked 52 minutes ago -
If the K b of a weak base is 6.3 × 10 − 6 , what...
asked 58 minutes ago -
Which of the following is the minimum amount of moles of NaOH
that must be added...
asked 1 hour ago
 Predict the products of the acid-base reaction between the following substances: Interactive 3D display mode Draw the molecules or ions on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced Template toolbars, including charges where needed. The single bond is active by default.
Predict the products of the acid-base reaction between the following substances: Interactive 3D display mode Draw the molecules or ions on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced Template toolbars, including charges where needed. The single bond is active by default.
 Draw the products of the acid-base reaction between the following species: Interactive 3D display mode Draw the products on the canvas by choosing buttons from the Tools (for bonds), Atoms
Draw the products of the acid-base reaction between the following species: Interactive 3D display mode Draw the products on the canvas by choosing buttons from the Tools (for bonds), Atoms
 Predict the products of the acid-base reaction between the
following substances:
*Draw* the molecules or ions.
Predict the products of the acid-base reaction between the following substances: Draw the molecules or ions.
Predict the products of the acid-base reaction between the
following substances:
*Draw* the molecules or ions.
Predict the products of the acid-base reaction between the following substances: Draw the molecules or ions.
 Part A Predict the products of the acid-base reaction between the following substances: Interactive MarvinView
Part A Predict the products of the acid-base reaction between the following substances: Interactive MarvinView
 Draw the keto tautomeric form of the following compounds:
Part A Draw the keto tautomeric form of the following compound Interactive 3D display mode H3C CH2 ÓH
Draw the keto tautomeric form of the following compounds:
Part A Draw the keto tautomeric form of the following compound Interactive 3D display mode H3C CH2 ÓH
 Draw the organic product of the acid-base reaction between the following species: Interactive 3D display mode Draw the molecule or ion on the canvas by choosing buttons from the Tools (for bonds), Atoms and Advanced Template toolbars, including charges where needed. The single bond is active b default.
Draw the organic product of the acid-base reaction between the following species: Interactive 3D display mode Draw the molecule or ion on the canvas by choosing buttons from the Tools (for bonds), Atoms and Advanced Template toolbars, including charges where needed. The single bond is active b default.
 Please Help!
Draw the major product of the following reaction: Interactive 3D display mode HN CI
Please Help!
Draw the major product of the following reaction: Interactive 3D display mode HN CI
 yeAgtiCicHDafV2XDloDXmVc7271oBw7QYban Investments waigreens O SIMPLE REACTIONS Predicting the products of a neutralization reaction Predict the products of the reaction below. That is, complete the right-hand side of the chemical equation. Be sure your equation is balanced. HBrCa(OH), ? X
yeAgtiCicHDafV2XDloDXmVc7271oBw7QYban Investments waigreens O SIMPLE REACTIONS Predicting the products of a neutralization reaction Predict the products of the reaction below. That is, complete the right-hand side of the chemical equation. Be sure your equation is balanced. HBrCa(OH), ? X
 Predict the products that could form. Next to the products you
draw, right either “products not favored” or “products
favored”
4. This question continues on the next page. For each of the following acid-base reactions: (0.3 pts ea = 3.0 pts) a. Predict the products that could form. b. Next to the products you draw, write either "Products not favored" or "Products favored." f. HCECH + CH3Li →
Predict the products that could form. Next to the products you
draw, right either “products not favored” or “products
favored”
4. This question continues on the next page. For each of the following acid-base reactions: (0.3 pts ea = 3.0 pts) a. Predict the products that could form. b. Next to the products you draw, write either "Products not favored" or "Products favored." f. HCECH + CH3Li →
 Draw all possible products formed when the compound shown below
undergoes reaction with 1 equivalent of Br2.
Interactive 3D display mode
Draw all possible products formed when the compound shown below
undergoes reaction with 1 equivalent of Br2.
Interactive 3D display mode