What is the product produced in the following reaction? In the following acid catalyzed dehydration reaction,...
What is the product produced in the following reaction?
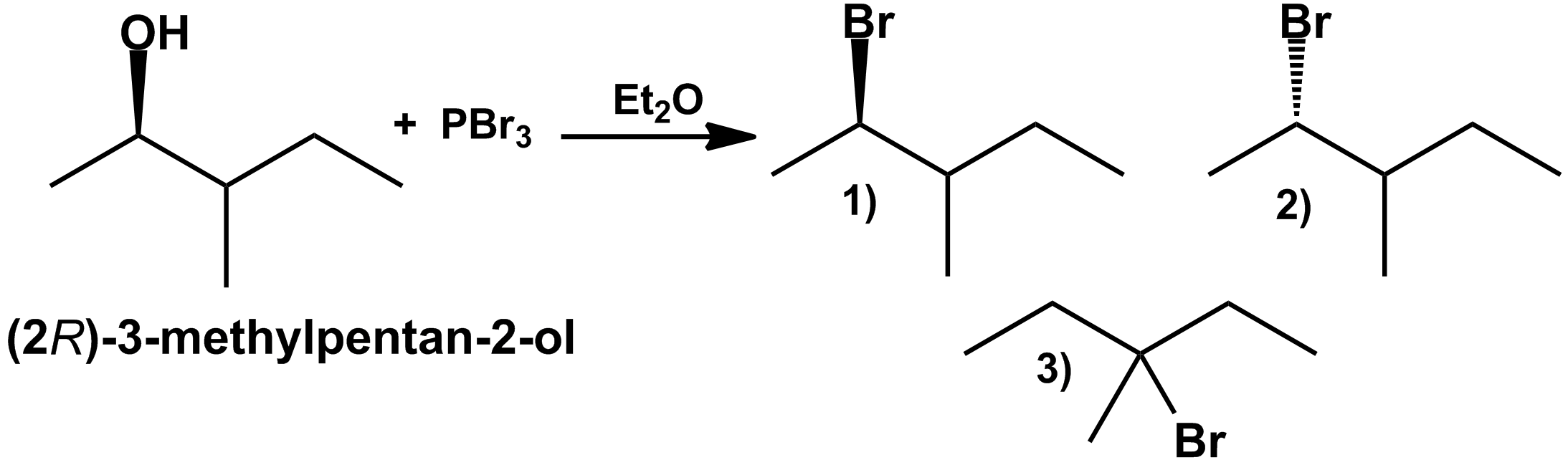
In the following acid catalyzed dehydration reaction, what is the alcohol that is most likely to give an alkene with a rearranged carbon skeleton? (Hint: a hydride shift does not rearrange the carbon skeleton)

Arrange the given alcohols in order of reactivity in an acid-catalyzed dehydration reaction.

Homework Answers

option 3 is most likely to give an alkene with a rearranged carbon skeleton.
Explanation: structure 3 involves a methyl shift and gives rearranged carbon skeleton as product.
order of reactivity in an acid-catalyzed dehydration reaction: 2 > 3 > 1
Explanation: Acid catalyzed dehydration involves formation of carbocation as an intermediate. tertiary carbocation is more stable so structure 2 gives faster dehydration reaction.
Add Answer to:
What is the product produced in the following reaction? In the following acid catalyzed dehydration reaction,...
Arrange the following alcohols according to increasing rates of their acid-catalyzed dehydration to an alkene (slowest...
 Arrange the following alcohols according to increasing rates of
their acid-catalyzed dehydration to an alkene (slowest rate at
top).
Arrange the following alcohols according to increasing rates of their acid-catalyzed dehydration to an alkene (slowest rate at top).
Arrange the following alcohols according to increasing rates of
their acid-catalyzed dehydration to an alkene (slowest rate at
top).
Arrange the following alcohols according to increasing rates of their acid-catalyzed dehydration to an alkene (slowest rate at top).
how does the mixture of isomeric alkenes produced by an acid-catalyzed dehydration reaction compare with the...
 how does the mixture of isomeric alkenes produced by an
acid-catalyzed dehydration reaction compare with the alkene mixture
from a base-catalyzed dehydrochlorination reaction? Are either or
both sets or products influenced primarily by product
stability?
E1/E2 Elimination Reactions. Acid-Catalyzed Dehydration of 2-Me Base-Catalyzed Dehydrochlorination of 2-Chloro-2-methylbutane Acid-Catalyzed Dehydration of 2-Methyl-2-butanol and Adapted for an experiment published in More J.R - Hammond, C.N. Schatz, PF: Morrill, T.C.: Modern Projects and P. Hammond CN: Schatz, P.F., Morrill... Experiments in Organic Chemistry, 2nd...
how does the mixture of isomeric alkenes produced by an
acid-catalyzed dehydration reaction compare with the alkene mixture
from a base-catalyzed dehydrochlorination reaction? Are either or
both sets or products influenced primarily by product
stability?
E1/E2 Elimination Reactions. Acid-Catalyzed Dehydration of 2-Me Base-Catalyzed Dehydrochlorination of 2-Chloro-2-methylbutane Acid-Catalyzed Dehydration of 2-Methyl-2-butanol and Adapted for an experiment published in More J.R - Hammond, C.N. Schatz, PF: Morrill, T.C.: Modern Projects and P. Hammond CN: Schatz, P.F., Morrill... Experiments in Organic Chemistry, 2nd...
HELP 12. Which of the following is/are the major products of reaction below) co > e) 13. The acid catalyzed dehy...
 HELP
12. Which of the following is/are the major products of reaction below) co > e) 13. The acid catalyzed dehydration of the alcohol below gives a major product which results from a carbocation rearrangement. Identify the major product. H2504) heat 14. What is the intermediate in the following reaction? D + Bra CH2Cle> 9) ol) 15. a) b) 2) d) The hydrohalogenation reaction can be described as: Anti-Markovnikov addition of halogen to an alkene Anti-Markovnikov addition of HX to...
HELP
12. Which of the following is/are the major products of reaction below) co > e) 13. The acid catalyzed dehydration of the alcohol below gives a major product which results from a carbocation rearrangement. Identify the major product. H2504) heat 14. What is the intermediate in the following reaction? D + Bra CH2Cle> 9) ol) 15. a) b) 2) d) The hydrohalogenation reaction can be described as: Anti-Markovnikov addition of halogen to an alkene Anti-Markovnikov addition of HX to...
Arrange the following alcohols according to increasing rates of their acid-catalyzed dehydration to an alkene (slowest...
 Arrange the following alcohols according to increasing rates of their acid-catalyzed dehydration to an alkene (slowest rate at top).
Arrange the following alcohols according to increasing rates of their acid-catalyzed dehydration to an alkene (slowest rate at top).
Chemistry 2401 Experiment 7 Acid Catalyzed Dehydration of 3.3-Dimethyl-2-Butanol with Rearrangement Experiment 7 Pre-lab Assignment Dehydration...
 Chemistry 2401 Experiment 7 Acid Catalyzed Dehydration of 3.3-Dimethyl-2-Butanol with Rearrangement Experiment 7 Pre-lab Assignment Dehydration of 3,3-Dimethyl-2-Butanol 1. Draw the structures of all the expected products from the following reactions: a) 3-methyl-3-pentanol + H = b)2-methylcyclohexanol + H - c) 2-methyl-2-butanol + H - d) 2,2-dimethylcyclohexanol + H = This reaction will give a complex mixture of products, since rearrangements can occur. Show the full mechanism for this reaction and how the possible products can form from the carbocation...
Chemistry 2401 Experiment 7 Acid Catalyzed Dehydration of 3.3-Dimethyl-2-Butanol with Rearrangement Experiment 7 Pre-lab Assignment Dehydration of 3,3-Dimethyl-2-Butanol 1. Draw the structures of all the expected products from the following reactions: a) 3-methyl-3-pentanol + H = b)2-methylcyclohexanol + H - c) 2-methyl-2-butanol + H - d) 2,2-dimethylcyclohexanol + H = This reaction will give a complex mixture of products, since rearrangements can occur. Show the full mechanism for this reaction and how the possible products can form from the carbocation...
product(s) would be produced by acid-catalyzed dehydration of the alcohol? CH CH3CCH2CH2 CH3 HA, heat -H20)...
 product(s) would be produced by acid-catalyzed dehydration of the alcohol? CH CH3CCH2CH2 CH3 HA, heat -H20) он A) CH B) C) CH3 D) CHa E) CH3CCH2CH2CH3 CH2-CCH2CH2CH3 and CH3C-CHCH2CH3 CH3CHCH CHCH3 and CH3CHCH2 CH3CHCH2CH-СНГ CH3 CH3 CH3 CH3 CH3 CHa CH3 11. Which alkene would you expect to be the major product of the following dehydration? so, /heol A) I B) II C) II D) IV E) V Page 6
product(s) would be produced by acid-catalyzed dehydration of the alcohol? CH CH3CCH2CH2 CH3 HA, heat -H20) он A) CH B) C) CH3 D) CHa E) CH3CCH2CH2CH3 CH2-CCH2CH2CH3 and CH3C-CHCH2CH3 CH3CHCH CHCH3 and CH3CHCH2 CH3CHCH2CH-СНГ CH3 CH3 CH3 CH3 CH3 CHa CH3 11. Which alkene would you expect to be the major product of the following dehydration? so, /heol A) I B) II C) II D) IV E) V Page 6
Acid-catalyzed addition of water to an alkene yields an alcohol with Markovnikov regiochemistry
 Acid-catalyzed addition of water to an alkene yields an alcohol
with Markovnikov regiochemistry. The electrophilic H+ adds to the
sp^2 carbon with the most hydrogens to yield the most stable
carbocation intermediate, which then adds water to give the product
alcohol. To avoid the possibility of rearrangement and still give a Markovnikov alcohol, alkenes can instead be treated with mercury(II) acetate in aqucous THF and then subsequently reduced with sodium borohydride. This reaction proceeds through a cyclic mercurinium ion intermediate which...
Acid-catalyzed addition of water to an alkene yields an alcohol
with Markovnikov regiochemistry. The electrophilic H+ adds to the
sp^2 carbon with the most hydrogens to yield the most stable
carbocation intermediate, which then adds water to give the product
alcohol. To avoid the possibility of rearrangement and still give a Markovnikov alcohol, alkenes can instead be treated with mercury(II) acetate in aqucous THF and then subsequently reduced with sodium borohydride. This reaction proceeds through a cyclic mercurinium ion intermediate which...
can somebody explain this. how we get the product plzz 311 7.11 CARBOCATION STABILITY PRACTICE PROBLEM...
 can somebody explain this. how we get the product
plzz
311 7.11 CARBOCATION STABILITY PRACTICE PROBLEM 7.18 When the compound called isoborneol is heated with 9 M sulfuric acid, the product of the reaction is the compound called camphene and not bornylene, as one might expect. Using models to assist you, write a step-by-step mechanism showing how camphene is formed. HO H,O+ not heat Bornylene Camphene Isoborneol 7.11B Rearrangement After Dehydration of a Primary Alcohol Rearrangements also accompany the dehydration...
can somebody explain this. how we get the product
plzz
311 7.11 CARBOCATION STABILITY PRACTICE PROBLEM 7.18 When the compound called isoborneol is heated with 9 M sulfuric acid, the product of the reaction is the compound called camphene and not bornylene, as one might expect. Using models to assist you, write a step-by-step mechanism showing how camphene is formed. HO H,O+ not heat Bornylene Camphene Isoborneol 7.11B Rearrangement After Dehydration of a Primary Alcohol Rearrangements also accompany the dehydration...
Practice: Show the product and mechanism for the hydration of cyclohexene in a reaction catalyzed by...
 Practice: Show the product and mechanism for the hydration of cyclohexene in a reaction catalyzed by sulfuric acid. A similar reaction can be used to make ethers (instead of alcohols) using an alcohol (instead of water). This reaction is called alcoholysis of an alkene. Practice: Draw the product and mechanism for the alcoholysis of 2-methylpropene and methanol with sulfuric acid.
Practice: Show the product and mechanism for the hydration of cyclohexene in a reaction catalyzed by sulfuric acid. A similar reaction can be used to make ethers (instead of alcohols) using an alcohol (instead of water). This reaction is called alcoholysis of an alkene. Practice: Draw the product and mechanism for the alcoholysis of 2-methylpropene and methanol with sulfuric acid.
please help The dehydration of alcohols Under acidic conditions, alcohols will undergo a dehydration reaction, producing...
 please help
The dehydration of alcohols Under acidic conditions, alcohols will undergo a dehydration reaction, producing an alkene and water. During the reaction, the hydroxyl group of the alcohol and a hydrogen atom from an adjacent carbon atom are lost. The reaction takes place in three steps, as shown in the diagram. (Figure 1) In the first step, a hydrogen lon bonds to the hydroxyl group of the alcohol. In the second step, the protonated hydroxl group leaves the compound,...
please help
The dehydration of alcohols Under acidic conditions, alcohols will undergo a dehydration reaction, producing an alkene and water. During the reaction, the hydroxyl group of the alcohol and a hydrogen atom from an adjacent carbon atom are lost. The reaction takes place in three steps, as shown in the diagram. (Figure 1) In the first step, a hydrogen lon bonds to the hydroxyl group of the alcohol. In the second step, the protonated hydroxl group leaves the compound,...
Most questions answered within 3 hours.
-
Based on the range, which of the following sets of scores has
the greatest variability? 3,...
asked 1 hour ago -
Ripples in a pond travel at a velocity of 3 m/s with one peak
passing a...
asked 54 minutes ago -
A man stands on the roof of a building of height 13.0 mm and
throws a...
asked 1 hour ago -
The extent to which assets are financed by borrowed funds and
other liabilities is indicated by:...
asked 2 hours ago -
Explain in detail
Germany is the fifth largest economy
explain what goods and services Germany specializes...
asked 2 hours ago -
The density of platinum is 21.45 g/mL. If a cube of platinum
with a mass of...
asked 2 hours ago -
Accounts Receivable
Sales
A/R Posting
Extended Sales Invoice
Packing Slip
Compare invoice to packing slip 2...
asked 2 hours ago -
Michaella, age 23, is a full-time law student and is claimed by
her parents as a...
asked 2 hours ago -
Why are polymers not typically casted into products?
asked 2 hours ago -
When rolling a die 129 times, what is the probability of rolling
a 6 no more...
asked 2 hours ago -
4. A call option currently sells for $7.75. It has a strike
price of $85 and...
asked 2 hours ago -
1.
You need to prepare 10.0 liters of an acid aqueous solution with a
pH of...
asked 2 hours ago
 Arrange the following alcohols according to increasing rates of
their acid-catalyzed dehydration to an alkene (slowest rate at
top).
Arrange the following alcohols according to increasing rates of their acid-catalyzed dehydration to an alkene (slowest rate at top).
Arrange the following alcohols according to increasing rates of
their acid-catalyzed dehydration to an alkene (slowest rate at
top).
Arrange the following alcohols according to increasing rates of their acid-catalyzed dehydration to an alkene (slowest rate at top).
 how does the mixture of isomeric alkenes produced by an
acid-catalyzed dehydration reaction compare with the alkene mixture
from a base-catalyzed dehydrochlorination reaction? Are either or
both sets or products influenced primarily by product
stability?
E1/E2 Elimination Reactions. Acid-Catalyzed Dehydration of 2-Me Base-Catalyzed Dehydrochlorination of 2-Chloro-2-methylbutane Acid-Catalyzed Dehydration of 2-Methyl-2-butanol and Adapted for an experiment published in More J.R - Hammond, C.N. Schatz, PF: Morrill, T.C.: Modern Projects and P. Hammond CN: Schatz, P.F., Morrill... Experiments in Organic Chemistry, 2nd...
how does the mixture of isomeric alkenes produced by an
acid-catalyzed dehydration reaction compare with the alkene mixture
from a base-catalyzed dehydrochlorination reaction? Are either or
both sets or products influenced primarily by product
stability?
E1/E2 Elimination Reactions. Acid-Catalyzed Dehydration of 2-Me Base-Catalyzed Dehydrochlorination of 2-Chloro-2-methylbutane Acid-Catalyzed Dehydration of 2-Methyl-2-butanol and Adapted for an experiment published in More J.R - Hammond, C.N. Schatz, PF: Morrill, T.C.: Modern Projects and P. Hammond CN: Schatz, P.F., Morrill... Experiments in Organic Chemistry, 2nd...
 HELP
12. Which of the following is/are the major products of reaction below) co > e) 13. The acid catalyzed dehydration of the alcohol below gives a major product which results from a carbocation rearrangement. Identify the major product. H2504) heat 14. What is the intermediate in the following reaction? D + Bra CH2Cle> 9) ol) 15. a) b) 2) d) The hydrohalogenation reaction can be described as: Anti-Markovnikov addition of halogen to an alkene Anti-Markovnikov addition of HX to...
HELP
12. Which of the following is/are the major products of reaction below) co > e) 13. The acid catalyzed dehydration of the alcohol below gives a major product which results from a carbocation rearrangement. Identify the major product. H2504) heat 14. What is the intermediate in the following reaction? D + Bra CH2Cle> 9) ol) 15. a) b) 2) d) The hydrohalogenation reaction can be described as: Anti-Markovnikov addition of halogen to an alkene Anti-Markovnikov addition of HX to...
 Arrange the following alcohols according to increasing rates of their acid-catalyzed dehydration to an alkene (slowest rate at top).
Arrange the following alcohols according to increasing rates of their acid-catalyzed dehydration to an alkene (slowest rate at top).
 Chemistry 2401 Experiment 7 Acid Catalyzed Dehydration of 3.3-Dimethyl-2-Butanol with Rearrangement Experiment 7 Pre-lab Assignment Dehydration of 3,3-Dimethyl-2-Butanol 1. Draw the structures of all the expected products from the following reactions: a) 3-methyl-3-pentanol + H = b)2-methylcyclohexanol + H - c) 2-methyl-2-butanol + H - d) 2,2-dimethylcyclohexanol + H = This reaction will give a complex mixture of products, since rearrangements can occur. Show the full mechanism for this reaction and how the possible products can form from the carbocation...
Chemistry 2401 Experiment 7 Acid Catalyzed Dehydration of 3.3-Dimethyl-2-Butanol with Rearrangement Experiment 7 Pre-lab Assignment Dehydration of 3,3-Dimethyl-2-Butanol 1. Draw the structures of all the expected products from the following reactions: a) 3-methyl-3-pentanol + H = b)2-methylcyclohexanol + H - c) 2-methyl-2-butanol + H - d) 2,2-dimethylcyclohexanol + H = This reaction will give a complex mixture of products, since rearrangements can occur. Show the full mechanism for this reaction and how the possible products can form from the carbocation...
 product(s) would be produced by acid-catalyzed dehydration of the alcohol? CH CH3CCH2CH2 CH3 HA, heat -H20) он A) CH B) C) CH3 D) CHa E) CH3CCH2CH2CH3 CH2-CCH2CH2CH3 and CH3C-CHCH2CH3 CH3CHCH CHCH3 and CH3CHCH2 CH3CHCH2CH-СНГ CH3 CH3 CH3 CH3 CH3 CHa CH3 11. Which alkene would you expect to be the major product of the following dehydration? so, /heol A) I B) II C) II D) IV E) V Page 6
product(s) would be produced by acid-catalyzed dehydration of the alcohol? CH CH3CCH2CH2 CH3 HA, heat -H20) он A) CH B) C) CH3 D) CHa E) CH3CCH2CH2CH3 CH2-CCH2CH2CH3 and CH3C-CHCH2CH3 CH3CHCH CHCH3 and CH3CHCH2 CH3CHCH2CH-СНГ CH3 CH3 CH3 CH3 CH3 CHa CH3 11. Which alkene would you expect to be the major product of the following dehydration? so, /heol A) I B) II C) II D) IV E) V Page 6
 can somebody explain this. how we get the product
plzz
311 7.11 CARBOCATION STABILITY PRACTICE PROBLEM 7.18 When the compound called isoborneol is heated with 9 M sulfuric acid, the product of the reaction is the compound called camphene and not bornylene, as one might expect. Using models to assist you, write a step-by-step mechanism showing how camphene is formed. HO H,O+ not heat Bornylene Camphene Isoborneol 7.11B Rearrangement After Dehydration of a Primary Alcohol Rearrangements also accompany the dehydration...
can somebody explain this. how we get the product
plzz
311 7.11 CARBOCATION STABILITY PRACTICE PROBLEM 7.18 When the compound called isoborneol is heated with 9 M sulfuric acid, the product of the reaction is the compound called camphene and not bornylene, as one might expect. Using models to assist you, write a step-by-step mechanism showing how camphene is formed. HO H,O+ not heat Bornylene Camphene Isoborneol 7.11B Rearrangement After Dehydration of a Primary Alcohol Rearrangements also accompany the dehydration...
 Practice: Show the product and mechanism for the hydration of cyclohexene in a reaction catalyzed by sulfuric acid. A similar reaction can be used to make ethers (instead of alcohols) using an alcohol (instead of water). This reaction is called alcoholysis of an alkene. Practice: Draw the product and mechanism for the alcoholysis of 2-methylpropene and methanol with sulfuric acid.
Practice: Show the product and mechanism for the hydration of cyclohexene in a reaction catalyzed by sulfuric acid. A similar reaction can be used to make ethers (instead of alcohols) using an alcohol (instead of water). This reaction is called alcoholysis of an alkene. Practice: Draw the product and mechanism for the alcoholysis of 2-methylpropene and methanol with sulfuric acid.
 please help
The dehydration of alcohols Under acidic conditions, alcohols will undergo a dehydration reaction, producing an alkene and water. During the reaction, the hydroxyl group of the alcohol and a hydrogen atom from an adjacent carbon atom are lost. The reaction takes place in three steps, as shown in the diagram. (Figure 1) In the first step, a hydrogen lon bonds to the hydroxyl group of the alcohol. In the second step, the protonated hydroxl group leaves the compound,...
please help
The dehydration of alcohols Under acidic conditions, alcohols will undergo a dehydration reaction, producing an alkene and water. During the reaction, the hydroxyl group of the alcohol and a hydrogen atom from an adjacent carbon atom are lost. The reaction takes place in three steps, as shown in the diagram. (Figure 1) In the first step, a hydrogen lon bonds to the hydroxyl group of the alcohol. In the second step, the protonated hydroxl group leaves the compound,...