Homework Answers

Add Answer to:
Using the table, calculate the difference in energy between the two conformers shown. *The cost of...
the table below shows the relationship between the difference in energy of two conformers of the same compound and the...
 the table below shows the relationship between the difference
in energy of two conformers of the same compound and the
percentages of the more stable
(2) The table below shows the relationship between the difference in energy (AG°) of two conformers of the same compound and the percentages of the more stable (A) and the less stable (B) conformers in their equilibrium mixture. OA (kJ/mol) more stable more stable AG oB 50 67 75 50 33 25 1.7 2.7 3.4...
the table below shows the relationship between the difference
in energy of two conformers of the same compound and the
percentages of the more stable
(2) The table below shows the relationship between the difference in energy (AG°) of two conformers of the same compound and the percentages of the more stable (A) and the less stable (B) conformers in their equilibrium mixture. OA (kJ/mol) more stable more stable AG oB 50 67 75 50 33 25 1.7 2.7 3.4...
Using the table below, calculate the difference in energy between a) the lowest energy conformations of...
Using the table below, calculate the difference in energy between a) the lowest energy conformations of cis- and trans-1,2-dimethylcyclohexane. b) the highest and lowest energy conformations of methyl cyclohexane, trans-1,2-dimethylcyclohexane and cis-1,3-dimethylcyclohexane Type of Strain Interaction Associated Energy Eclipsing of a pair of H's 4kj/mol Eclipsing of a H and a CH3 group 6kj/mol Eclipsing of a pair of CH3 groups 11kj/mol Gauche (60) Interactin between CH3 Groups 3.8 kj/mol 1,3 CH3 to H Interaction on Cyclohexane Chair 3.8kj/mol 1,3...
Part B Using the table on the left, calculate the total kilocalories for fat (rounding off...
 Part B
Using the table on the left, calculate the total kilocalories
for fat (rounding off the kilocalories to the tens place).
Express the energy to two significant figures and
include the appropriate units.
Part C Using the table on the left, calculate the total
kilocalories for protein (rounding off the kilocalories to the tens
place).
Express the energy to two significant figures and
include the appropriate units.
Part D
Determine the total kilocalories for the meal (rounding off to...
Part B
Using the table on the left, calculate the total kilocalories
for fat (rounding off the kilocalories to the tens place).
Express the energy to two significant figures and
include the appropriate units.
Part C Using the table on the left, calculate the total
kilocalories for protein (rounding off the kilocalories to the tens
place).
Express the energy to two significant figures and
include the appropriate units.
Part D
Determine the total kilocalories for the meal (rounding off to...
Calculate the energy difference (E) for the transition from n = 1 to n = 6...
Calculate the energy difference (E) for the transition from n = 1 to n = 6 energy levels of hydrogen per 1 mol of H atoms. (Report your answer to at least 3 significant figures.)
Part A Compute the energy separation between the ground and first excited states for an electron...
Part A Compute the energy separation between the ground and first excited states for an electron in a one-dimensional box that is 5.10 angstroms in length. Express the energy difference in kJ⋅mol−1. Express your answer to three significant figures and include the appropriate units. E= Part B Compute the wavelength of light (in nm) corresponding to this energy. Express your answer to three significant figures and include the appropriate units. λ=
Part A Calculate the amount of steric strain in H CH3 СН3 Express your answer using...
 Part A Calculate the amount of steric strain in H CH3 СН3 Express your answer using two significant figures. V AC mo ? a total strain energy = kcal/mol Submit Request Answer Review Constants 1 Per Part B Calculate the amount of steric strain in CH3 CH3 CH3 Express your answer using two significant figures. IVO AQ RO ? a total strain energy = kcal/mol Submit Request Answer Part C Which conformer would predominate at equilibrium? CH3 H ΤΗ O...
Part A Calculate the amount of steric strain in H CH3 СН3 Express your answer using two significant figures. V AC mo ? a total strain energy = kcal/mol Submit Request Answer Review Constants 1 Per Part B Calculate the amount of steric strain in CH3 CH3 CH3 Express your answer using two significant figures. IVO AQ RO ? a total strain energy = kcal/mol Submit Request Answer Part C Which conformer would predominate at equilibrium? CH3 H ΤΗ O...
Part A Compute the energy separation between the ground and second excited states for an electron in a one-dimensional b...
Part A Compute the energy separation between the ground and second excited states for an electron in a one-dimensional box that is 7.70 angstroms in length. Express the energy difference in kJ⋅mol−1. Express your answer to three significant figures and include the appropriate units. Part B Compute the wavelength of light (in nm) corresponding to this energy. Express your answer to three significant figures and include the appropriate units.
Bookmark A 0.28 kg block oscillates between two 18 N/m springs, as shown in the figure....
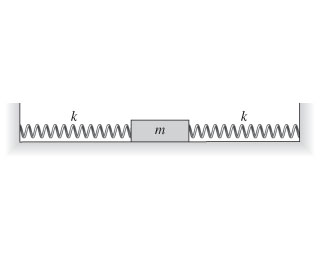 Bookmark A 0.28 kg block oscillates between two 18 N/m springs,
as shown in the figure. (Figure
https://media.cheggcdn.com/media/981/981295cb-0dd2-43e9-9dbb-73b2ec3d730b/phpwRyUKp.png
What's the oscillation period? Express your answer using two
significant figures. Compare with the period of the same block
oscillating on a single 18 N/m spring. Express your answer using
two significant figures.
Bookmark A 0.28 kg block oscillates between two 18 N/m springs,
as shown in the figure. (Figure
https://media.cheggcdn.com/media/981/981295cb-0dd2-43e9-9dbb-73b2ec3d730b/phpwRyUKp.png
What's the oscillation period? Express your answer using two
significant figures. Compare with the period of the same block
oscillating on a single 18 N/m spring. Express your answer using
two significant figures.
Calculate the bond energy per mole for breaking all the bonds in methane, CH4
Part ACalculate the bond energy per mole for breaking all the bonds in methane, CH4.Express your answer to four significant figures and include the appropriate units.ΔHCH4 =1656 kJmolCorrectIn CH4, the energy required to break one C−H bond is 414 kJ/mol. Since there are four C−H bonds in CH4, the energy ΔHCH4 for breaking all the bonds is calculated asΔHCH4=4×bond energy of C−H bond=4×414 kJ/mol=1656 kJ/mol CH4 moleculesPart BCalculate the bond energy per mole for breaking all the bonds of oxygen,...
Constants | Periodic Table Using values of Kb in Appendix C in the textbook, calculate values of Ka for each of the...
 Constants | Periodic Table
Using values of Kb in Appendix C in the textbook, calculate
values of Ka for each of the following ions:
Part A
Propylammonium ion, C3H7NH3+
Express your answer using two significant figures.
Part B
Hydroxylammonium ion, NH3OH+
Express your answer using two significant figures.
Part C
Anilinium ion, C6H5NH3+
Express your answer using two significant figures.
Part D
Pyridinium ion, C5H5NH+
Express your answer using two significant figures.
Constants | Periodic Table
Using values of Kb in Appendix C in the textbook, calculate
values of Ka for each of the following ions:
Part A
Propylammonium ion, C3H7NH3+
Express your answer using two significant figures.
Part B
Hydroxylammonium ion, NH3OH+
Express your answer using two significant figures.
Part C
Anilinium ion, C6H5NH3+
Express your answer using two significant figures.
Part D
Pyridinium ion, C5H5NH+
Express your answer using two significant figures.
Most questions answered within 3 hours.
-
Using Python. Write a function clean2(aList) that takes a list
of integers aList as argument, and...
asked 19 seconds ago -
Intrapreneurship is defined as the
development of an enterprise culture with an existing (Nandan,
2009). An...
asked 14 minutes ago -
QUESTION 1 Tamiflu is a drug used to treat influenza. A study
found that Tamiflu reduced...
asked 18 minutes ago -
Which of the following would be less likely to be used to
allocate factory overhead when...
asked 24 minutes ago -
Transverse pulses travel with a speed of 195 m/s along a taut
copper wire whose diameter...
asked 22 minutes ago -
Sampling for Engineers Homework question 11.51
Suppose that Y1;Y2;Y3;Y4 denote a random sample from a
Poisson(θ)...
asked 26 minutes ago -
Which of the following is cleaved by regulated intramembrane
proteolysis (RIP)? Select any/all answers that apply....
asked 25 minutes ago -
Random characters You decide to create a program characters.py
that fills a two-dimensional list with random...
asked 37 minutes ago -
What are the major sources of error in your determination of the
molar mass? Select all...
asked 41 minutes ago -
A. Suppose your manager indicates that for a normally
distributed data set you are analyzing, your...
asked 1 hour ago -
During the month of August, the average temperature of a lake
next to a local college...
asked 44 minutes ago -
Which of these four researchers is most likely to have made Type
1 error
Kim who...
asked 58 minutes ago

 the table below shows the relationship between the difference
in energy of two conformers of the same compound and the
percentages of the more stable
(2) The table below shows the relationship between the difference in energy (AG°) of two conformers of the same compound and the percentages of the more stable (A) and the less stable (B) conformers in their equilibrium mixture. OA (kJ/mol) more stable more stable AG oB 50 67 75 50 33 25 1.7 2.7 3.4...
the table below shows the relationship between the difference
in energy of two conformers of the same compound and the
percentages of the more stable
(2) The table below shows the relationship between the difference in energy (AG°) of two conformers of the same compound and the percentages of the more stable (A) and the less stable (B) conformers in their equilibrium mixture. OA (kJ/mol) more stable more stable AG oB 50 67 75 50 33 25 1.7 2.7 3.4...
 Part B
Using the table on the left, calculate the total kilocalories
for fat (rounding off the kilocalories to the tens place).
Express the energy to two significant figures and
include the appropriate units.
Part C Using the table on the left, calculate the total
kilocalories for protein (rounding off the kilocalories to the tens
place).
Express the energy to two significant figures and
include the appropriate units.
Part D
Determine the total kilocalories for the meal (rounding off to...
Part B
Using the table on the left, calculate the total kilocalories
for fat (rounding off the kilocalories to the tens place).
Express the energy to two significant figures and
include the appropriate units.
Part C Using the table on the left, calculate the total
kilocalories for protein (rounding off the kilocalories to the tens
place).
Express the energy to two significant figures and
include the appropriate units.
Part D
Determine the total kilocalories for the meal (rounding off to...
 Part A Calculate the amount of steric strain in H CH3 СН3 Express your answer using two significant figures. V AC mo ? a total strain energy = kcal/mol Submit Request Answer Review Constants 1 Per Part B Calculate the amount of steric strain in CH3 CH3 CH3 Express your answer using two significant figures. IVO AQ RO ? a total strain energy = kcal/mol Submit Request Answer Part C Which conformer would predominate at equilibrium? CH3 H ΤΗ O...
Part A Calculate the amount of steric strain in H CH3 СН3 Express your answer using two significant figures. V AC mo ? a total strain energy = kcal/mol Submit Request Answer Review Constants 1 Per Part B Calculate the amount of steric strain in CH3 CH3 CH3 Express your answer using two significant figures. IVO AQ RO ? a total strain energy = kcal/mol Submit Request Answer Part C Which conformer would predominate at equilibrium? CH3 H ΤΗ O...
 Constants | Periodic Table
Using values of Kb in Appendix C in the textbook, calculate
values of Ka for each of the following ions:
Part A
Propylammonium ion, C3H7NH3+
Express your answer using two significant figures.
Part B
Hydroxylammonium ion, NH3OH+
Express your answer using two significant figures.
Part C
Anilinium ion, C6H5NH3+
Express your answer using two significant figures.
Part D
Pyridinium ion, C5H5NH+
Express your answer using two significant figures.
Constants | Periodic Table
Using values of Kb in Appendix C in the textbook, calculate
values of Ka for each of the following ions:
Part A
Propylammonium ion, C3H7NH3+
Express your answer using two significant figures.
Part B
Hydroxylammonium ion, NH3OH+
Express your answer using two significant figures.
Part C
Anilinium ion, C6H5NH3+
Express your answer using two significant figures.
Part D
Pyridinium ion, C5H5NH+
Express your answer using two significant figures.