What is the mechanism for base-catalyzed transesterification with methanol where the ester reacts...
What is the mechanism for base-catalyzed transesterification with methanol where the ester reacts with an alcohol solvent to form a new ester that contains the alcohol's OR group?
Please draw out mechanism and explain. the reaction will use the reagents XS NaOCH3 (from NaOH, CH3OH) to convert vegatable oil (a fatty acid triester) to glycerol and 3 moles of methyl oleate (biodiesel)
Homework Answers
Answer: Trans-esterification is just a reaction of exchanging the alkoxy (-OR) group of an ester with the new alkoxy group which usually comes from the solvent as a result of the reaction of base with the solvent. To show the mechanism, a simple ester has been taken and the reaction of trans-esterification is illustrated with arrows. In the base-catalyzed trans-esterification, the first step is the formation of Na-OR' in which the alkoxide (-OR') is from the solvent. As methanol is the solvent in our case. It will be sodium methoxide. In the second step, the formed -OCH3 will attack the electrophilic carbonyl carbon of the ester and will eliminate the -OR which was already present in the ester. This process is known as trans-esterification.
Trans-esterification of vegetable oil with NaOH and Methanol is drawn at the end. Please refer to the attached image.

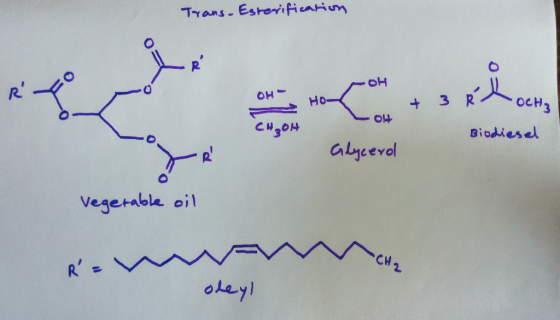
Add Answer to:
What is the mechanism for base-catalyzed transesterification with methanol where the ester reacts...
Draw the mechanism for the base-catalyzed transesterification of a vegetable oil using methanol
Draw the mechanism for the base-catalyzed transesterification of a vegetable oil using methanol
What would be my percent yield if i retrieved 3.1020 grams of soap? thank you, please show all work.
 what would be my percent yield if i retrieved 3.1020 grams of
soap? thank you, please show all work.
SAPONIFICATION (HYDROLYSIS OF A FAD Fats and edible oils consitute a special class of esters called riglycerides in which alcohol part of the molecule, the trihydroxy compound glycerol, forms with three molecules of various long-chain carboxylic acids (called fatty acids). Ty reactions of simple esters can also be carried out on fats and oils,i acid-or-base-catalyzed hydrolysis. three ester linkages carboxylic acids...
what would be my percent yield if i retrieved 3.1020 grams of
soap? thank you, please show all work.
SAPONIFICATION (HYDROLYSIS OF A FAD Fats and edible oils consitute a special class of esters called riglycerides in which alcohol part of the molecule, the trihydroxy compound glycerol, forms with three molecules of various long-chain carboxylic acids (called fatty acids). Ty reactions of simple esters can also be carried out on fats and oils,i acid-or-base-catalyzed hydrolysis. three ester linkages carboxylic acids...
Questions and Exercises 1. In the course of reflux of the triglyceride in a sodium hydroxide solu...
 Organic Chemistry Help!
Questions and Exercises 1. In the course of reflux of the triglyceride in a sodium hydroxide solution you will notice a lot of foaming. Explain. What is left behind in the aqueous layer after filtration of the saponification product? The starting material and the product of the saponification reaction have similar melting points. How do you know you actually isolated a new product rather than just recovered the starting material? Give at least two different methods to...
Organic Chemistry Help!
Questions and Exercises 1. In the course of reflux of the triglyceride in a sodium hydroxide solution you will notice a lot of foaming. Explain. What is left behind in the aqueous layer after filtration of the saponification product? The starting material and the product of the saponification reaction have similar melting points. How do you know you actually isolated a new product rather than just recovered the starting material? Give at least two different methods to...
PREPARATION OF PHARMACEUTICALS - Fischer Esterification Reactions Q: Calculate the expected mass of both methyl salicylate and aspirin assuming 100% yield. EXPERIMENT7 PREPARATION OF PHARMACEUTICALS...
 PREPARATION OF PHARMACEUTICALS - Fischer Esterification
Reactions
Q: Calculate the expected mass of both methyl salicylate and
aspirin assuming 100% yield.
EXPERIMENT7 PREPARATION OF PHARMACEUTICALS Fischer Esterification Reactions Almost 2500 years ago, physicians such as Hippocrates recommended that patients chew on the bark of the willow to alleviate pain. The active ingredient in willow bark was found to be salicin, a compound made of a molecule of salicyl alcohol bonded to a p-D-glucose molecule. In the stomach, the bond between...
PREPARATION OF PHARMACEUTICALS - Fischer Esterification
Reactions
Q: Calculate the expected mass of both methyl salicylate and
aspirin assuming 100% yield.
EXPERIMENT7 PREPARATION OF PHARMACEUTICALS Fischer Esterification Reactions Almost 2500 years ago, physicians such as Hippocrates recommended that patients chew on the bark of the willow to alleviate pain. The active ingredient in willow bark was found to be salicin, a compound made of a molecule of salicyl alcohol bonded to a p-D-glucose molecule. In the stomach, the bond between...
Consider the SN2 reaction of butyl bromide with hydroxide ion , Assuming no other changes, what...
Consider the SN2 reaction of butyl bromide with hydroxide ion , Assuming no other changes, what effect on the reaction rate would result from simultaneously doubling the concentrations of both butyl bromide and hydroxyl ion No effect (B)It would double the rate (C)it would increase the rate six times (D) It would increase the rate four times. 2. Which alky halide would you expect to undergo SN1 hydrolysis most rapidly? (A) (CH3)3C-I (B) (CH3)3C-Br (C) (CH3)3C-Cl (D) (CH3)3C-F 3....
What is the theoretical yield (in grams) and percent yield of your triphenylmethanol product in this...
 What is the theoretical yield (in grams) and percent yield of your
triphenylmethanol product in this experiment? (Weight of
triphenylmethanol: 0.060g)
BACKGROUND AND THEORY The Grignard reaction was one of the first organometallic reactions discovered and is still one of the most useful synthetically. By reacting an organohalide (usually a bromide) with magnesium in ethereal solvent, carbon becomes a nucleophile. Grignard reagents are the starting points for the syntheses of many alkanes, primary, secondary, and tertiary alcohols, alkenes, and carboxylic...
What is the theoretical yield (in grams) and percent yield of your
triphenylmethanol product in this experiment? (Weight of
triphenylmethanol: 0.060g)
BACKGROUND AND THEORY The Grignard reaction was one of the first organometallic reactions discovered and is still one of the most useful synthetically. By reacting an organohalide (usually a bromide) with magnesium in ethereal solvent, carbon becomes a nucleophile. Grignard reagents are the starting points for the syntheses of many alkanes, primary, secondary, and tertiary alcohols, alkenes, and carboxylic...
Most questions answered within 3 hours.
-
Kylie is a single mom with two dependent children,
Tanner, age 7 and Olivia, age 11....
asked 41 minutes ago -
Phosphorous + bromine = phosphorous tribromide. If 35.0 g of
bromine are reacted and 27.9 grams...
asked 2 hours ago -
Derive the long wavelength limit of the Planck energy density
distribution
asked 1 hour ago -
Calculate the pH of each of the following solutions.
0.50 M HBr
3.1×10−4 M KOH
4.2×10−5...
asked 5 hours ago -
For the year ended December 31, Depot Max’s cost of merchandise
sold was $85,600. Inventory at the...
asked 5 hours ago -
Week 10 - Professional Memo Assignment
Professional Memo Assignment
Your mission for this week, should you...
asked 5 hours ago -
Write a Python program that stores the data for each
player on the team, and it...
asked 5 hours ago -
In
the last 3 months, mike never knows when he is going to get his
allowance...
asked 6 hours ago -
Is Ca(OH)2 a Bronsted base, Lewis base, or both? Why?
asked 6 hours ago -
1A- Why don’t voters complain about U.S. tariffs on imported
sugar?
Because sugar is only a...
asked 6 hours ago -
Cash Payback Period
Primera Banco is evaluating two capital investment proposals for
a drive-up ATM kiosk,...
asked 6 hours ago -
Create a button in Swift (Xcode) that will create a charge,
create a charge using Stripe's...
asked 6 hours ago
 what would be my percent yield if i retrieved 3.1020 grams of
soap? thank you, please show all work.
SAPONIFICATION (HYDROLYSIS OF A FAD Fats and edible oils consitute a special class of esters called riglycerides in which alcohol part of the molecule, the trihydroxy compound glycerol, forms with three molecules of various long-chain carboxylic acids (called fatty acids). Ty reactions of simple esters can also be carried out on fats and oils,i acid-or-base-catalyzed hydrolysis. three ester linkages carboxylic acids...
what would be my percent yield if i retrieved 3.1020 grams of
soap? thank you, please show all work.
SAPONIFICATION (HYDROLYSIS OF A FAD Fats and edible oils consitute a special class of esters called riglycerides in which alcohol part of the molecule, the trihydroxy compound glycerol, forms with three molecules of various long-chain carboxylic acids (called fatty acids). Ty reactions of simple esters can also be carried out on fats and oils,i acid-or-base-catalyzed hydrolysis. three ester linkages carboxylic acids...
 Organic Chemistry Help!
Questions and Exercises 1. In the course of reflux of the triglyceride in a sodium hydroxide solution you will notice a lot of foaming. Explain. What is left behind in the aqueous layer after filtration of the saponification product? The starting material and the product of the saponification reaction have similar melting points. How do you know you actually isolated a new product rather than just recovered the starting material? Give at least two different methods to...
Organic Chemistry Help!
Questions and Exercises 1. In the course of reflux of the triglyceride in a sodium hydroxide solution you will notice a lot of foaming. Explain. What is left behind in the aqueous layer after filtration of the saponification product? The starting material and the product of the saponification reaction have similar melting points. How do you know you actually isolated a new product rather than just recovered the starting material? Give at least two different methods to...
 PREPARATION OF PHARMACEUTICALS - Fischer Esterification
Reactions
Q: Calculate the expected mass of both methyl salicylate and
aspirin assuming 100% yield.
EXPERIMENT7 PREPARATION OF PHARMACEUTICALS Fischer Esterification Reactions Almost 2500 years ago, physicians such as Hippocrates recommended that patients chew on the bark of the willow to alleviate pain. The active ingredient in willow bark was found to be salicin, a compound made of a molecule of salicyl alcohol bonded to a p-D-glucose molecule. In the stomach, the bond between...
PREPARATION OF PHARMACEUTICALS - Fischer Esterification
Reactions
Q: Calculate the expected mass of both methyl salicylate and
aspirin assuming 100% yield.
EXPERIMENT7 PREPARATION OF PHARMACEUTICALS Fischer Esterification Reactions Almost 2500 years ago, physicians such as Hippocrates recommended that patients chew on the bark of the willow to alleviate pain. The active ingredient in willow bark was found to be salicin, a compound made of a molecule of salicyl alcohol bonded to a p-D-glucose molecule. In the stomach, the bond between...
 What is the theoretical yield (in grams) and percent yield of your
triphenylmethanol product in this experiment? (Weight of
triphenylmethanol: 0.060g)
BACKGROUND AND THEORY The Grignard reaction was one of the first organometallic reactions discovered and is still one of the most useful synthetically. By reacting an organohalide (usually a bromide) with magnesium in ethereal solvent, carbon becomes a nucleophile. Grignard reagents are the starting points for the syntheses of many alkanes, primary, secondary, and tertiary alcohols, alkenes, and carboxylic...
What is the theoretical yield (in grams) and percent yield of your
triphenylmethanol product in this experiment? (Weight of
triphenylmethanol: 0.060g)
BACKGROUND AND THEORY The Grignard reaction was one of the first organometallic reactions discovered and is still one of the most useful synthetically. By reacting an organohalide (usually a bromide) with magnesium in ethereal solvent, carbon becomes a nucleophile. Grignard reagents are the starting points for the syntheses of many alkanes, primary, secondary, and tertiary alcohols, alkenes, and carboxylic...