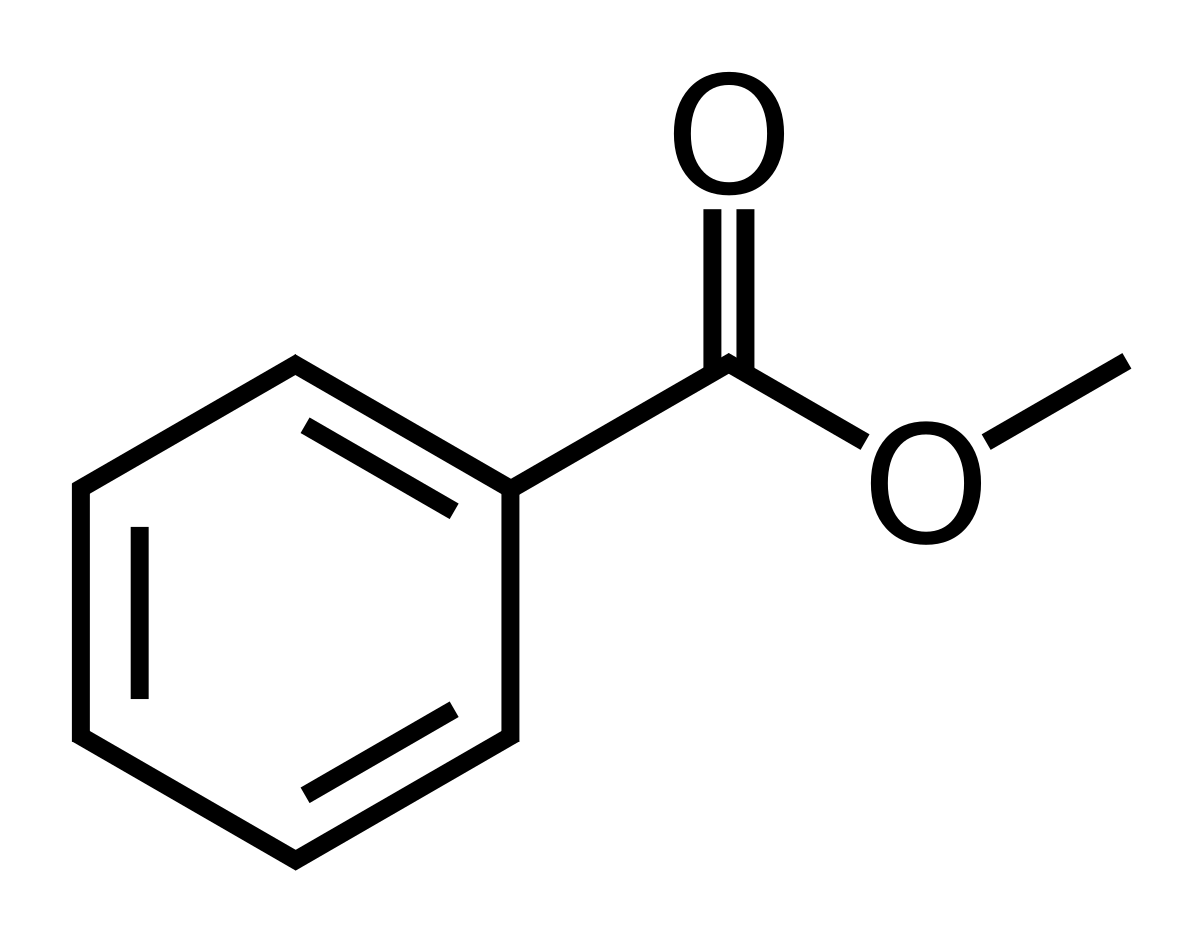
I need help with the assignment there are 4 1. 3.9ppm, singlet, 3H 2. 7.4ppm, triplet, 2H 3. 7.6ppm, triplet, 1H 4. 8.1p...
I need help with the assignment there are 4
1. 3.9ppm, singlet, 3H
2. 7.4ppm, triplet, 2H
3. 7.6ppm, triplet, 1H
4. 8.1ppm, doublet, 2H
Homework Answers


Add Answer to:
I need help with the assignment there are 4 1. 3.9ppm, singlet, 3H 2. 7.4ppm, triplet, 2H 3. 7.6ppm, triplet, 1H 4. 8.1p...
I need help determing the multiplicity (singlet, doublet, triplet, etc.) and the expected integration (number of...
 I need help determing the multiplicity (singlet, doublet,
triplet, etc.) and the expected integration (number of protons- eg.
1H, 2H, 3H) for each spectra.
OH Source: www.chemicalbook.com Br. 2,46-tribromophenol H-NMR Spectrum 7 6 2 p-nitroacetanilide H-NMR Spectrum 2,1 pm 8.2ppm lo.uppm ヒ-7. 8ppm 11 10 9 87 65 4 3 2 1 c-CH
I need help determing the multiplicity (singlet, doublet,
triplet, etc.) and the expected integration (number of protons- eg.
1H, 2H, 3H) for each spectra.
OH Source: www.chemicalbook.com Br. 2,46-tribromophenol H-NMR Spectrum 7 6 2 p-nitroacetanilide H-NMR Spectrum 2,1 pm 8.2ppm lo.uppm ヒ-7. 8ppm 11 10 9 87 65 4 3 2 1 c-CH
Draw the structure that would be responsible or the following spectra: 5. CH.0;septet (1H), singlet (3H),...
 Draw the structure that would be responsible or the following
spectra:
5. CH.0;septet (1H), singlet (3H), doublet (6H) 11 10 8 7 6 4 3 2 1 0 5 ppm 6. C1H1402, multiplet (5H), singlet (2H), triplet (2H), multiplet (2H), triplet (3H) E 11 10 9 2 ppm
Draw the structure that would be responsible or the following
spectra:
5. CH.0;septet (1H), singlet (3H), doublet (6H) 11 10 8 7 6 4 3 2 1 0 5 ppm 6. C1H1402, multiplet (5H), singlet (2H), triplet (2H), multiplet (2H), triplet (3H) E 11 10 9 2 ppm
Which compound matches the following 1H NMR spectrum? doublet, 2H singlet 3H doublet 2H br. singlet...
 Which compound matches the following 1H NMR spectrum? doublet, 2H singlet 3H doublet 2H br. singlet 1H 10 PPM NO2 OH OH A D OD ОА OC B
Which compound matches the following 1H NMR spectrum? doublet, 2H singlet 3H doublet 2H br. singlet 1H 10 PPM NO2 OH OH A D OD ОА OC B
Deduce the final structure consistent to the 1H-NMR data. C10H12O2 2.0ppm(singlet, 3H), 2.9 ppm (triplet, 2H), 4.3 ppm (...
Deduce the final structure consistent to the 1H-NMR data. C10H12O2 2.0ppm(singlet, 3H), 2.9 ppm (triplet, 2H), 4.3 ppm (triplet, 2H)7.3 ppm (singlet, 5H)
please help! c 3.86 ppm, singlet, 3H d-2.52 ppm, singlet, 3H Compound A: CoH1002 TSI a...
 please help!
c 3.86 ppm, singlet, 3H d-2.52 ppm, singlet, 3H Compound A: CoH1002 TSI a 7.89 ppm, doublet, 2H b-7.89 ppm, doublet, 2H Unsat. Index- (2C+2-H-X+N)/2 8, ppm 9.0 7.0 60 3.0 2.0 ? 3, 5-dimethylbenzaldehyde, NaOH, H2O, heat h 2.31 ppm, singlet, 6H 3-1 a #8 ppm, doublet, 2H b-7.60 ppm, doublet, 1H c- 7.33 ppm, singlet, 2H Compound B: C1H1O2 g-3.86 ppm, singlet, 3H d-7.28 ppm, doublet, 1 H e 6.99 ppm, singlet, 1H C f 6.98...
please help!
c 3.86 ppm, singlet, 3H d-2.52 ppm, singlet, 3H Compound A: CoH1002 TSI a 7.89 ppm, doublet, 2H b-7.89 ppm, doublet, 2H Unsat. Index- (2C+2-H-X+N)/2 8, ppm 9.0 7.0 60 3.0 2.0 ? 3, 5-dimethylbenzaldehyde, NaOH, H2O, heat h 2.31 ppm, singlet, 6H 3-1 a #8 ppm, doublet, 2H b-7.60 ppm, doublet, 1H c- 7.33 ppm, singlet, 2H Compound B: C1H1O2 g-3.86 ppm, singlet, 3H d-7.28 ppm, doublet, 1 H e 6.99 ppm, singlet, 1H C f 6.98...
5) Chemical Formula: CgHN IR: Two weak peaks at 3350cm-1 3H Doublet, IH Triplet, 1H Triplet,...
 5) Chemical Formula: CgHN IR: Two weak peaks at 3350cm-1 3H Doublet, IH Triplet, 1H Triplet, 1H Doublet, 1H 2H 4 2 PPM 6) Chemical Formula: C10H1202 IR: strong peak 1750cm-1 PPM
5) Chemical Formula: CgHN IR: Two weak peaks at 3350cm-1 3H Doublet, IH Triplet, 1H Triplet, 1H Doublet, 1H 2H 4 2 PPM 6) Chemical Formula: C10H1202 IR: strong peak 1750cm-1 PPM
Having trouble figuring out these two unknown compounds. 17) Formula: C8H10O with a strong peak a...
 Having trouble figuring out
these two unknown compounds. 17) Formula: C8H10O with a strong peak
at 3200 cm^-1
18) Formula: C10H13Br I just need to know the structures of each
of them if you could please help me out!
17) Chemical Formulac: CHroO IR: strong broad peak at 3200 cm-1 Singlet, 3 Triplet, 1H Singlet, 2hH Doublet, 1H Singlet, 1H Singlet, 1H Doublet, 1H 0 2 6 PPM 18) Chemical Formula: C10H13Br Doublet, 3H Doublet, 2H Triplet, 3hH Quartet, 2H...
Having trouble figuring out
these two unknown compounds. 17) Formula: C8H10O with a strong peak
at 3200 cm^-1
18) Formula: C10H13Br I just need to know the structures of each
of them if you could please help me out!
17) Chemical Formulac: CHroO IR: strong broad peak at 3200 cm-1 Singlet, 3 Triplet, 1H Singlet, 2hH Doublet, 1H Singlet, 1H Singlet, 1H Doublet, 1H 0 2 6 PPM 18) Chemical Formula: C10H13Br Doublet, 3H Doublet, 2H Triplet, 3hH Quartet, 2H...
C9H12 13C NMR 7 peaks 1H NMR δ 1.13 (triplet, 3H); δ 1.71 (multiplet, 2H); δ...
C9H12 13C NMR 7 peaks 1H NMR δ 1.13 (triplet, 3H); δ 1.71 (multiplet, 2H); δ 2.64 (triplet, 2H); δ 7.34 (multiplet, 5H) Degree of Unsaturation _______ Draw the structure of your compound and indicate the ppm of each of the H’s. 2. C5H10O2 13C NMR 5 peaks 1H NMR δ 0.93 (triplet, 3H); δ 1.70 (multiplet, 2H); δ 2.25 (triplet, 2H); δ 3.59 (singlet, 3H) Degree of Unsaturation _______ Draw the structure of your compound and indicate the ppm...
3. Propose structures for compounds that fit the following 1H NMR data: a. C.H.Cl2: 8 1.60...
 3. Propose structures for compounds that fit the following 1H NMR data: a. C.H.Cl2: 8 1.60 (doublet, 3H), 2.15 (multiplet, 2H), 3.72 (triplet, 2H), and 4.27 (multiplet, 1H) b. C.H.Br: 8 1.1 (doublet, 6H), 1.9 (multiplet, 1H), and 3.4 (doublet, 2H) c. CH 40:8 0.9 (triplet, 6H), 1.6 (sextet, 4H), and 2.4 (triplet, 4H) d. CsH1002: 8 1.2 (doublet, 6H), 2.0 (singlet, 3H) and 5.0 (septet, 1H)
3. Propose structures for compounds that fit the following 1H NMR data: a. C.H.Cl2: 8 1.60 (doublet, 3H), 2.15 (multiplet, 2H), 3.72 (triplet, 2H), and 4.27 (multiplet, 1H) b. C.H.Br: 8 1.1 (doublet, 6H), 1.9 (multiplet, 1H), and 3.4 (doublet, 2H) c. CH 40:8 0.9 (triplet, 6H), 1.6 (sextet, 4H), and 2.4 (triplet, 4H) d. CsH1002: 8 1.2 (doublet, 6H), 2.0 (singlet, 3H) and 5.0 (septet, 1H)
3. Propose structures for compounds that fit the following 1H NMR data: a. C4H2Cl2: 8 1.60...
 3. Propose structures for compounds that fit the following 1H NMR data: a. C4H2Cl2: 8 1.60 (doublet, 3H), 2.15 (multiplet, 2H), 3.72 (triplet, 2H), and 4.27 (multiplet, 1H) b. C4H,Br: 8 1.1 (doublet, 6H), 1.9 (multiplet, 1H), and 3.4 (doublet, 2H) c. C-H140:8 0.9 (triplet, 6H), 1.6 (sextet, 4H), and 2.4 (triplet, 4H) d. C5H1002:8 1.2 (doublet, 6H), 2.0 (singlet, 3H) and 5.0 (septet, 1H)
3. Propose structures for compounds that fit the following 1H NMR data: a. C4H2Cl2: 8 1.60 (doublet, 3H), 2.15 (multiplet, 2H), 3.72 (triplet, 2H), and 4.27 (multiplet, 1H) b. C4H,Br: 8 1.1 (doublet, 6H), 1.9 (multiplet, 1H), and 3.4 (doublet, 2H) c. C-H140:8 0.9 (triplet, 6H), 1.6 (sextet, 4H), and 2.4 (triplet, 4H) d. C5H1002:8 1.2 (doublet, 6H), 2.0 (singlet, 3H) and 5.0 (septet, 1H)
Most questions answered within 3 hours.
-
The fuel economy of a 2011 Lexus RX 350 2wd 6 cylinder 3.5 L
automatic 5...
asked 1 second from now -
i.
the synthesized compund 2-bromo-butanal i have a IR peak at about
3200 and one at...
asked 3 seconds from now -
A
752 mL sample of water was placed in a 1000 gram pan of aluminum.
The...
asked 11 minutes ago -
1.In the context of chelation, what does binding strength mean?
What happens at the molecular level...
asked 4 minutes ago -
Describe two obstacles that makes fixing atmospheric nitrogen
difficult.
asked 23 minutes ago -
Evelyn incorporates her sole proprietorship, transferring it to
newly formed Papaya Corporation. The assets transferred have...
asked 9 minutes ago -
Assume that in a hydrogen atom, the electron circles the nucleus
in a circle of radius...
asked 18 minutes ago -
1 point) Given the significance level α=0.01 find the following:
(a) left-tailed z value z= (b)...
asked 12 minutes ago -
Calculate the expected value, the variance, and the standard
deviation of the given random variable X....
asked 14 minutes ago -
T
F 53) Most differences
between human groups are the result of biology rather than
culture....
asked 27 minutes ago -
A 5.20 mW helium neon laser emits a visible laser beam with a
wavelength of 633...
asked 30 minutes ago -
Assignment:
Your
organization has made a strategic decision
to
outsourcework
currently performed in house. You have...
asked 29 minutes ago
 I need help determing the multiplicity (singlet, doublet,
triplet, etc.) and the expected integration (number of protons- eg.
1H, 2H, 3H) for each spectra.
OH Source: www.chemicalbook.com Br. 2,46-tribromophenol H-NMR Spectrum 7 6 2 p-nitroacetanilide H-NMR Spectrum 2,1 pm 8.2ppm lo.uppm ヒ-7. 8ppm 11 10 9 87 65 4 3 2 1 c-CH
I need help determing the multiplicity (singlet, doublet,
triplet, etc.) and the expected integration (number of protons- eg.
1H, 2H, 3H) for each spectra.
OH Source: www.chemicalbook.com Br. 2,46-tribromophenol H-NMR Spectrum 7 6 2 p-nitroacetanilide H-NMR Spectrum 2,1 pm 8.2ppm lo.uppm ヒ-7. 8ppm 11 10 9 87 65 4 3 2 1 c-CH
 Draw the structure that would be responsible or the following
spectra:
5. CH.0;septet (1H), singlet (3H), doublet (6H) 11 10 8 7 6 4 3 2 1 0 5 ppm 6. C1H1402, multiplet (5H), singlet (2H), triplet (2H), multiplet (2H), triplet (3H) E 11 10 9 2 ppm
Draw the structure that would be responsible or the following
spectra:
5. CH.0;septet (1H), singlet (3H), doublet (6H) 11 10 8 7 6 4 3 2 1 0 5 ppm 6. C1H1402, multiplet (5H), singlet (2H), triplet (2H), multiplet (2H), triplet (3H) E 11 10 9 2 ppm
 Which compound matches the following 1H NMR spectrum? doublet, 2H singlet 3H doublet 2H br. singlet 1H 10 PPM NO2 OH OH A D OD ОА OC B
Which compound matches the following 1H NMR spectrum? doublet, 2H singlet 3H doublet 2H br. singlet 1H 10 PPM NO2 OH OH A D OD ОА OC B
 please help!
c 3.86 ppm, singlet, 3H d-2.52 ppm, singlet, 3H Compound A: CoH1002 TSI a 7.89 ppm, doublet, 2H b-7.89 ppm, doublet, 2H Unsat. Index- (2C+2-H-X+N)/2 8, ppm 9.0 7.0 60 3.0 2.0 ? 3, 5-dimethylbenzaldehyde, NaOH, H2O, heat h 2.31 ppm, singlet, 6H 3-1 a #8 ppm, doublet, 2H b-7.60 ppm, doublet, 1H c- 7.33 ppm, singlet, 2H Compound B: C1H1O2 g-3.86 ppm, singlet, 3H d-7.28 ppm, doublet, 1 H e 6.99 ppm, singlet, 1H C f 6.98...
please help!
c 3.86 ppm, singlet, 3H d-2.52 ppm, singlet, 3H Compound A: CoH1002 TSI a 7.89 ppm, doublet, 2H b-7.89 ppm, doublet, 2H Unsat. Index- (2C+2-H-X+N)/2 8, ppm 9.0 7.0 60 3.0 2.0 ? 3, 5-dimethylbenzaldehyde, NaOH, H2O, heat h 2.31 ppm, singlet, 6H 3-1 a #8 ppm, doublet, 2H b-7.60 ppm, doublet, 1H c- 7.33 ppm, singlet, 2H Compound B: C1H1O2 g-3.86 ppm, singlet, 3H d-7.28 ppm, doublet, 1 H e 6.99 ppm, singlet, 1H C f 6.98...
 5) Chemical Formula: CgHN IR: Two weak peaks at 3350cm-1 3H Doublet, IH Triplet, 1H Triplet, 1H Doublet, 1H 2H 4 2 PPM 6) Chemical Formula: C10H1202 IR: strong peak 1750cm-1 PPM
5) Chemical Formula: CgHN IR: Two weak peaks at 3350cm-1 3H Doublet, IH Triplet, 1H Triplet, 1H Doublet, 1H 2H 4 2 PPM 6) Chemical Formula: C10H1202 IR: strong peak 1750cm-1 PPM
 Having trouble figuring out
these two unknown compounds. 17) Formula: C8H10O with a strong peak
at 3200 cm^-1
18) Formula: C10H13Br I just need to know the structures of each
of them if you could please help me out!
17) Chemical Formulac: CHroO IR: strong broad peak at 3200 cm-1 Singlet, 3 Triplet, 1H Singlet, 2hH Doublet, 1H Singlet, 1H Singlet, 1H Doublet, 1H 0 2 6 PPM 18) Chemical Formula: C10H13Br Doublet, 3H Doublet, 2H Triplet, 3hH Quartet, 2H...
Having trouble figuring out
these two unknown compounds. 17) Formula: C8H10O with a strong peak
at 3200 cm^-1
18) Formula: C10H13Br I just need to know the structures of each
of them if you could please help me out!
17) Chemical Formulac: CHroO IR: strong broad peak at 3200 cm-1 Singlet, 3 Triplet, 1H Singlet, 2hH Doublet, 1H Singlet, 1H Singlet, 1H Doublet, 1H 0 2 6 PPM 18) Chemical Formula: C10H13Br Doublet, 3H Doublet, 2H Triplet, 3hH Quartet, 2H...
 3. Propose structures for compounds that fit the following 1H NMR data: a. C.H.Cl2: 8 1.60 (doublet, 3H), 2.15 (multiplet, 2H), 3.72 (triplet, 2H), and 4.27 (multiplet, 1H) b. C.H.Br: 8 1.1 (doublet, 6H), 1.9 (multiplet, 1H), and 3.4 (doublet, 2H) c. CH 40:8 0.9 (triplet, 6H), 1.6 (sextet, 4H), and 2.4 (triplet, 4H) d. CsH1002: 8 1.2 (doublet, 6H), 2.0 (singlet, 3H) and 5.0 (septet, 1H)
3. Propose structures for compounds that fit the following 1H NMR data: a. C.H.Cl2: 8 1.60 (doublet, 3H), 2.15 (multiplet, 2H), 3.72 (triplet, 2H), and 4.27 (multiplet, 1H) b. C.H.Br: 8 1.1 (doublet, 6H), 1.9 (multiplet, 1H), and 3.4 (doublet, 2H) c. CH 40:8 0.9 (triplet, 6H), 1.6 (sextet, 4H), and 2.4 (triplet, 4H) d. CsH1002: 8 1.2 (doublet, 6H), 2.0 (singlet, 3H) and 5.0 (septet, 1H)
 3. Propose structures for compounds that fit the following 1H NMR data: a. C4H2Cl2: 8 1.60 (doublet, 3H), 2.15 (multiplet, 2H), 3.72 (triplet, 2H), and 4.27 (multiplet, 1H) b. C4H,Br: 8 1.1 (doublet, 6H), 1.9 (multiplet, 1H), and 3.4 (doublet, 2H) c. C-H140:8 0.9 (triplet, 6H), 1.6 (sextet, 4H), and 2.4 (triplet, 4H) d. C5H1002:8 1.2 (doublet, 6H), 2.0 (singlet, 3H) and 5.0 (septet, 1H)
3. Propose structures for compounds that fit the following 1H NMR data: a. C4H2Cl2: 8 1.60 (doublet, 3H), 2.15 (multiplet, 2H), 3.72 (triplet, 2H), and 4.27 (multiplet, 1H) b. C4H,Br: 8 1.1 (doublet, 6H), 1.9 (multiplet, 1H), and 3.4 (doublet, 2H) c. C-H140:8 0.9 (triplet, 6H), 1.6 (sextet, 4H), and 2.4 (triplet, 4H) d. C5H1002:8 1.2 (doublet, 6H), 2.0 (singlet, 3H) and 5.0 (septet, 1H)