
Pyrrole undergoes nitration by electrophilic aromatic substitution. Complete the mechanism by drawing curved arrows, the structure of the charged intermediate, and the structure of the major uncharged product. Omit electron lone pairs and bases.
Homework Answers
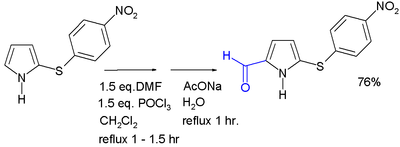
Pyrrole undergoes electrophilic aromatic substitution predominantly at the 2 and 5 positions. Two such reactions that are especially significant for producing functionalized pyrroles are theMannich reaction and the Vilsmeier-Haack reaction (depicted below), both of which are compatible with a variety of pyrrole substrates
Add Answer to:
Pyrrole undergoes nitration by electrophilic aromatic substitution. Complete the mechanism by drawing curved arrows, the structure...
Pyridine reacts with hydroxide by nucleophilic aromatic substitution. Complete the mechanism by drawing curved arrows, the...
 Pyridine reacts with hydroxide by nucleophilic aromatic
substitution. Complete the mechanism by drawing curved arrows, the
structure of the charged intermediate, and the structure of the
major uncharged product.
Violation
Pyridine reacts with hydroxide by nucleophilic aromatic
substitution. Complete the mechanism by drawing curved arrows, the
structure of the charged intermediate, and the structure of the
major uncharged product.
Violation
The electrophilic aromatic substitution of isopropylbenzene with FeBr3, Br2 gives 1-bromo-4-isopropylbenzene. Complete the curved-arrow mechanism below,...
 The electrophilic aromatic substitution of isopropylbenzene with
FeBr3, Br2 gives 1-bromo-4-isopropylbenzene. Complete the
curved-arrow mechanism below, beginning with formation of the
active brominating reagent. Remember to include lone pairs and
formal charges where appropriate.
The electrophilic aromatic substitution of isopropylbenzene with FeBr3, Br2 gives 1-bromo-4-isopropylbenzern Complete the curved-arrow mechanism below, beginning with formation of the active brominating reagent. Remember to include lone pairs and formal charges where appropriate. Draw the pro Overall transformation (ungraded) bromine and FeBr3 Include curved arrow(s)....
The electrophilic aromatic substitution of isopropylbenzene with
FeBr3, Br2 gives 1-bromo-4-isopropylbenzene. Complete the
curved-arrow mechanism below, beginning with formation of the
active brominating reagent. Remember to include lone pairs and
formal charges where appropriate.
The electrophilic aromatic substitution of isopropylbenzene with FeBr3, Br2 gives 1-bromo-4-isopropylbenzern Complete the curved-arrow mechanism below, beginning with formation of the active brominating reagent. Remember to include lone pairs and formal charges where appropriate. Draw the pro Overall transformation (ungraded) bromine and FeBr3 Include curved arrow(s)....
Complete the mechanism for the generation of the electrophile used for chlorination in electrophilic aromatic substitution....
 Complete the mechanism for the generation of the electrophile used for chlorination in electrophilic aromatic substitution. Add curved arrows, bonds, electron pairs, and charges where indicated. Add a curved arrow. Complete the structure and highlight the electrophilic chlorine atom. 0 8— A : ci : - -
Complete the mechanism for the generation of the electrophile used for chlorination in electrophilic aromatic substitution. Add curved arrows, bonds, electron pairs, and charges where indicated. Add a curved arrow. Complete the structure and highlight the electrophilic chlorine atom. 0 8— A : ci : - -
Complete the mechanism for generation of the electrophile used for bromination in electrophilic aromatic substitution. Add...
 Complete the mechanism for generation of the electrophile used
for bromination in electrophilic aromatic substitution. Add curved
arrows, bonds, electron pairs, and charges where indicated.
Complete the mechanism for generation of the electrophile used
for bromination in electrophilic aromatic substitution. Add curved
arrows, bonds, electron pairs, and charges where indicated.
Complete the mechanism for the electrophilic aromatic substitution of trifluorobenzene. Do not bother placing all of...
 Complete the mechanism for the electrophilic aromatic substitution of trifluorobenzene. Do not bother placing all of the lone pairs of electrons on the F atoms, but be sure to place them on all other atoms as needed. Do not draw curved arrows between the resonance structures.
Complete the mechanism for the electrophilic aromatic substitution of trifluorobenzene. Do not bother placing all of the lone pairs of electrons on the F atoms, but be sure to place them on all other atoms as needed. Do not draw curved arrows between the resonance structures.
When naphthalene undergoes an irreversible electrophilic aromatic substitution, the major product is the kinetic product, which...
 When naphthalene undergoes an irreversible electrophilic aromatic substitution, the major product is the kinetic product, which proceeds through the most stable arenium ion intermediate. With this in mind, draw the curved arrow mechanism for the first step of the electrophilic aromatic substitution of naphthalene with l^+ generated from l_2 and CuCl_2. If you predicted the correct regiochemistry in the first step, then you can draw the curved arrows and a resonance structure in step 2 with an unbroken benzenoid ring....
When naphthalene undergoes an irreversible electrophilic aromatic substitution, the major product is the kinetic product, which proceeds through the most stable arenium ion intermediate. With this in mind, draw the curved arrow mechanism for the first step of the electrophilic aromatic substitution of naphthalene with l^+ generated from l_2 and CuCl_2. If you predicted the correct regiochemistry in the first step, then you can draw the curved arrows and a resonance structure in step 2 with an unbroken benzenoid ring....
The electrophilic aromatic substitution of anisole with Br2 gives 4-bromoanisole. Complete the curved-arrow mechanism below; remember...
 The electrophilic aromatic substitution of anisole with Br2
gives 4-bromoanisole. Complete the curved-arrow mechanism below;
remember to include lone pairs and formal charges where
appropriate.
The electrophilic aromatic substitution of anisole with Br2
gives 4-bromoanisole. Complete the curved-arrow mechanism below;
remember to include lone pairs and formal charges where
appropriate.
Provide a complete mechanism for the above Electrophilic Aromatic Substitution Reaction. Pay close attention to details,...
 Provide a complete mechanism for the above Electrophilic Aromatic Substitution Reaction. Pay close attention to details, including lone pairs, formal charges and the use of curved arrows. B) Why is toluene an ortho/para director? Explain, using drawings to support your answer.
Provide a complete mechanism for the above Electrophilic Aromatic Substitution Reaction. Pay close attention to details, including lone pairs, formal charges and the use of curved arrows. B) Why is toluene an ortho/para director? Explain, using drawings to support your answer.
a See page 1127 13 Question (3 points) When naphthalene undergoes an irreversible electrophilic aromatic substitution,...
 a See page 1127 13 Question (3 points) When naphthalene undergoes an irreversible electrophilic aromatic substitution, the major product is the kinetic product, which proceeds through the most stable arenium ion intermediate. With this in mind, draw the curved arrow mechanism for the first step of the electrophilic aromatic substitution of naphthalene with the acylium ion generated from acetyl chloride and AlClg, Then draw the curved arrows and a resonance structure in step 2 with an unbroken benzenoid ring.Draw all...
a See page 1127 13 Question (3 points) When naphthalene undergoes an irreversible electrophilic aromatic substitution, the major product is the kinetic product, which proceeds through the most stable arenium ion intermediate. With this in mind, draw the curved arrow mechanism for the first step of the electrophilic aromatic substitution of naphthalene with the acylium ion generated from acetyl chloride and AlClg, Then draw the curved arrows and a resonance structure in step 2 with an unbroken benzenoid ring.Draw all...
Complete the mechanism for this cyclization reaction by drawing the missing intermediate structure and adding curved...
 Complete the mechanism for this cyclization reaction by drawing
the missing intermediate structure and adding curved arrows. Show
all lone pairs and arrows in each step.
Complete the mechanism for this cyclization reaction by drawing
the missing intermediate structure and adding curved arrows. Show
all lone pairs and arrows in each step.
Most questions answered within 3 hours.
-
Calculate the pH of a solution that is prepared by dissolving
0.470 mol of hydrocyanic acid...
asked 10 minutes ago -
TABLE 12-11 The director of admissions at a state college is
interested in seeing if admissions...
asked 15 minutes ago -
Give regular expressions describing each of the following
regular languages over Σ = {0,1}:
{w :...
asked 16 minutes ago -
If carbon-14 has a decay half-life of 5730 years, what fraction
of the original carbon-14 would...
asked 19 minutes ago -
Question 5. A person is murdered on a deserted
island. There are only 10 individuals who...
asked 27 minutes ago -
The Nature 2014 The genome of a Late Pleistocene human from a
Clovis burial site in...
asked 26 minutes ago -
Which 6 pertinent capacity questions will you address
that will be useful as part of the...
asked 31 minutes ago -
Discuss the global manifestations of religious fundamentalism in
the 1980s. What did the fundamentalists have in...
asked 43 minutes ago -
Roberto Corporation was organized on January 1, 2018. The firm
was authorized to issue 86,000 shares...
asked 47 minutes ago -
On December 31, 2018, Sage’s securities portfolio consisted of
the following common stocks. Security Quantity Cost...
asked 54 minutes ago -
topic:- integrated Enterprise Systems
* no hand writing
*the answer must be unique not copied “plagiarized...
asked 56 minutes ago -
In the carbon cycle, combustion reactions produce atmospheric
oxygen.
True
False
The empirical formulas for H2O,...
asked 1 hour ago

 Pyridine reacts with hydroxide by nucleophilic aromatic
substitution. Complete the mechanism by drawing curved arrows, the
structure of the charged intermediate, and the structure of the
major uncharged product.
Violation
Pyridine reacts with hydroxide by nucleophilic aromatic
substitution. Complete the mechanism by drawing curved arrows, the
structure of the charged intermediate, and the structure of the
major uncharged product.
Violation
 The electrophilic aromatic substitution of isopropylbenzene with
FeBr3, Br2 gives 1-bromo-4-isopropylbenzene. Complete the
curved-arrow mechanism below, beginning with formation of the
active brominating reagent. Remember to include lone pairs and
formal charges where appropriate.
The electrophilic aromatic substitution of isopropylbenzene with FeBr3, Br2 gives 1-bromo-4-isopropylbenzern Complete the curved-arrow mechanism below, beginning with formation of the active brominating reagent. Remember to include lone pairs and formal charges where appropriate. Draw the pro Overall transformation (ungraded) bromine and FeBr3 Include curved arrow(s)....
The electrophilic aromatic substitution of isopropylbenzene with
FeBr3, Br2 gives 1-bromo-4-isopropylbenzene. Complete the
curved-arrow mechanism below, beginning with formation of the
active brominating reagent. Remember to include lone pairs and
formal charges where appropriate.
The electrophilic aromatic substitution of isopropylbenzene with FeBr3, Br2 gives 1-bromo-4-isopropylbenzern Complete the curved-arrow mechanism below, beginning with formation of the active brominating reagent. Remember to include lone pairs and formal charges where appropriate. Draw the pro Overall transformation (ungraded) bromine and FeBr3 Include curved arrow(s)....
 Complete the mechanism for the generation of the electrophile used for chlorination in electrophilic aromatic substitution. Add curved arrows, bonds, electron pairs, and charges where indicated. Add a curved arrow. Complete the structure and highlight the electrophilic chlorine atom. 0 8— A : ci : - -
Complete the mechanism for the generation of the electrophile used for chlorination in electrophilic aromatic substitution. Add curved arrows, bonds, electron pairs, and charges where indicated. Add a curved arrow. Complete the structure and highlight the electrophilic chlorine atom. 0 8— A : ci : - -
 Complete the mechanism for generation of the electrophile used
for bromination in electrophilic aromatic substitution. Add curved
arrows, bonds, electron pairs, and charges where indicated.
Complete the mechanism for generation of the electrophile used
for bromination in electrophilic aromatic substitution. Add curved
arrows, bonds, electron pairs, and charges where indicated.
 Complete the mechanism for the electrophilic aromatic substitution of trifluorobenzene. Do not bother placing all of the lone pairs of electrons on the F atoms, but be sure to place them on all other atoms as needed. Do not draw curved arrows between the resonance structures.
Complete the mechanism for the electrophilic aromatic substitution of trifluorobenzene. Do not bother placing all of the lone pairs of electrons on the F atoms, but be sure to place them on all other atoms as needed. Do not draw curved arrows between the resonance structures.
 When naphthalene undergoes an irreversible electrophilic aromatic substitution, the major product is the kinetic product, which proceeds through the most stable arenium ion intermediate. With this in mind, draw the curved arrow mechanism for the first step of the electrophilic aromatic substitution of naphthalene with l^+ generated from l_2 and CuCl_2. If you predicted the correct regiochemistry in the first step, then you can draw the curved arrows and a resonance structure in step 2 with an unbroken benzenoid ring....
When naphthalene undergoes an irreversible electrophilic aromatic substitution, the major product is the kinetic product, which proceeds through the most stable arenium ion intermediate. With this in mind, draw the curved arrow mechanism for the first step of the electrophilic aromatic substitution of naphthalene with l^+ generated from l_2 and CuCl_2. If you predicted the correct regiochemistry in the first step, then you can draw the curved arrows and a resonance structure in step 2 with an unbroken benzenoid ring....
 The electrophilic aromatic substitution of anisole with Br2
gives 4-bromoanisole. Complete the curved-arrow mechanism below;
remember to include lone pairs and formal charges where
appropriate.
The electrophilic aromatic substitution of anisole with Br2
gives 4-bromoanisole. Complete the curved-arrow mechanism below;
remember to include lone pairs and formal charges where
appropriate.
 Provide a complete mechanism for the above Electrophilic Aromatic Substitution Reaction. Pay close attention to details, including lone pairs, formal charges and the use of curved arrows. B) Why is toluene an ortho/para director? Explain, using drawings to support your answer.
Provide a complete mechanism for the above Electrophilic Aromatic Substitution Reaction. Pay close attention to details, including lone pairs, formal charges and the use of curved arrows. B) Why is toluene an ortho/para director? Explain, using drawings to support your answer.
 a See page 1127 13 Question (3 points) When naphthalene undergoes an irreversible electrophilic aromatic substitution, the major product is the kinetic product, which proceeds through the most stable arenium ion intermediate. With this in mind, draw the curved arrow mechanism for the first step of the electrophilic aromatic substitution of naphthalene with the acylium ion generated from acetyl chloride and AlClg, Then draw the curved arrows and a resonance structure in step 2 with an unbroken benzenoid ring.Draw all...
a See page 1127 13 Question (3 points) When naphthalene undergoes an irreversible electrophilic aromatic substitution, the major product is the kinetic product, which proceeds through the most stable arenium ion intermediate. With this in mind, draw the curved arrow mechanism for the first step of the electrophilic aromatic substitution of naphthalene with the acylium ion generated from acetyl chloride and AlClg, Then draw the curved arrows and a resonance structure in step 2 with an unbroken benzenoid ring.Draw all...
 Complete the mechanism for this cyclization reaction by drawing
the missing intermediate structure and adding curved arrows. Show
all lone pairs and arrows in each step.
Complete the mechanism for this cyclization reaction by drawing
the missing intermediate structure and adding curved arrows. Show
all lone pairs and arrows in each step.