
Homework Answers
Request Answer!
We need at least 10 more requests to produce the answer.
0 / 10 have requested this problem solution
The more requests, the faster the answer.
Add Answer to:
please show work. i need to know how to do this for a
test
Question 18...
please show work i need to know this for a test Question 20 1 pts The...
 please show work i need to know this for a test
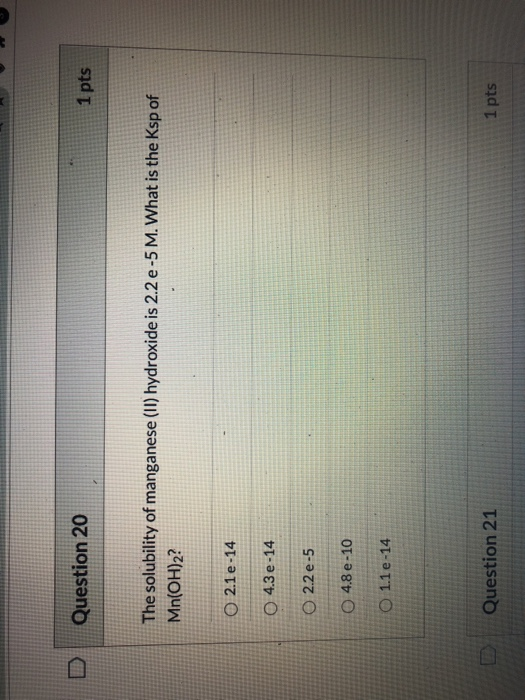
Question 20 1 pts The solubility of manganese (II) hydroxide is 2.2 e-5 M. What is the Ksp of Mn(OH)2? 0 2.1e-14 4.3 e-14 O 2.2 e-5 O 4.8 e -10 O 1.1 e -14 Question 21 1 pts
please show work i need to know this for a test
Question 20 1 pts The solubility of manganese (II) hydroxide is 2.2 e-5 M. What is the Ksp of Mn(OH)2? 0 2.1e-14 4.3 e-14 O 2.2 e-5 O 4.8 e -10 O 1.1 e -14 Question 21 1 pts
please show all work i need to know how to do this for a test The...
 please show all work i need to know how to do this for a
test
The reaction AB is first order in (A). Consider the following data. Time (s) 0.0 5.0 10.0 15.0 20.0 [A] (M) 0.20 0.14 0.10 0.071 0.050 The concentration of A is Mafter 40.0 s. 1.3 e-2 O 0.17 O 3.5 e-4 0 0.025 1.2
please show all work i need to know how to do this for a
test
The reaction AB is first order in (A). Consider the following data. Time (s) 0.0 5.0 10.0 15.0 20.0 [A] (M) 0.20 0.14 0.10 0.071 0.050 The concentration of A is Mafter 40.0 s. 1.3 e-2 O 0.17 O 3.5 e-4 0 0.025 1.2
need help ASAP 3 pts Question 2 Acetaminophen is a medication used to treat pain and...
 need help ASAP
3 pts Question 2 Acetaminophen is a medication used to treat pain and fever and contains Carbon, Hydrogen, Oxygen, and Nitrogen. Combustion analysis of a 15.0-mg sample of acetaminophen produced 8.10 mg of water. What is the percent by mass of Hydrogen in acetaminophen? 4.4% 12.0% 7.5% 3.0% 6.0% Question 4 1.5 pts AgCl, FeCO3. Hg Bry, and Pbly are compounds that are insoluble in water. Which compound would be more soluble in acidic solution? AgCl only...
need help ASAP
3 pts Question 2 Acetaminophen is a medication used to treat pain and fever and contains Carbon, Hydrogen, Oxygen, and Nitrogen. Combustion analysis of a 15.0-mg sample of acetaminophen produced 8.10 mg of water. What is the percent by mass of Hydrogen in acetaminophen? 4.4% 12.0% 7.5% 3.0% 6.0% Question 4 1.5 pts AgCl, FeCO3. Hg Bry, and Pbly are compounds that are insoluble in water. Which compound would be more soluble in acidic solution? AgCl only...
Question 3 7.5 pts Time Running Attempt due: Jur 1 Hour, 14 Mi The concentration of...
 Question 3 7.5 pts Time Running Attempt due: Jur 1 Hour, 14 Mi The concentration of Agt in a solution saturated with Ag2C2O4(s) is 1.50x10^-4 M. Calculate Ksp for AgzC204. Ksp = es
Question 3 7.5 pts Time Running Attempt due: Jur 1 Hour, 14 Mi The concentration of Agt in a solution saturated with Ag2C2O4(s) is 1.50x10^-4 M. Calculate Ksp for AgzC204. Ksp = es
please show work. i need to know how to do this for a test A flask...
 please show work. i need to know how to do this for a
test
A flask is charged with 0.124 mol of A and allowed to react to form B according to the reaction A(g) →B(g). The following data are obtained for [A] as the reaction proceeds: Time (s) 0.00 10.0- 20.0 30.0 40.0 Moles of A 0.124 0.110 0.088 0.073 0.054 The average rate of disappearance of A between 10 s and 20 s is mol/s. ect Answer 2.2...
please show work. i need to know how to do this for a
test
A flask is charged with 0.124 mol of A and allowed to react to form B according to the reaction A(g) →B(g). The following data are obtained for [A] as the reaction proceeds: Time (s) 0.00 10.0- 20.0 30.0 40.0 Moles of A 0.124 0.110 0.088 0.073 0.054 The average rate of disappearance of A between 10 s and 20 s is mol/s. ect Answer 2.2...
please show work. i need this for a test Question 21 1 pts The reaction AB...
 please show work. i need this for a test
Question 21 1 pts The reaction AB is first order in A. Consider the following data: Time (s) [A] (M) 0.0 2.3 10.0 0.58 20.0 0.14 The rate constant for this reaction is s-1 0.013 O 0.030 O 3.1e-3 O 0.14 O 3.0
please show work. i need this for a test
Question 21 1 pts The reaction AB is first order in A. Consider the following data: Time (s) [A] (M) 0.0 2.3 10.0 0.58 20.0 0.14 The rate constant for this reaction is s-1 0.013 O 0.030 O 3.1e-3 O 0.14 O 3.0
Please explain the correct answer for both questions. Thank you! Incorrect Question 7 0/1 pts The...
 Please explain the correct
answer for both questions. Thank you!
Incorrect Question 7 0/1 pts The concentration of iodide ions in a saturated solution of lead (11) iodide is_ The solubility product constant of Pblz is 1.4x10-8. 3.5x10-9 3.0x10-3 1.5x103 1.4x10-8 3.8x10-4 Incorrect Question 9 0/1 pts The molar solubility of lead () chloride (PbCl2) is 1.6x102 M. What is the Ksp of PbCl2? 4.1x10-6 5.0x10-4 1.6x10-2 3.1x107 1.6x10-5
Please explain the correct
answer for both questions. Thank you!
Incorrect Question 7 0/1 pts The concentration of iodide ions in a saturated solution of lead (11) iodide is_ The solubility product constant of Pblz is 1.4x10-8. 3.5x10-9 3.0x10-3 1.5x103 1.4x10-8 3.8x10-4 Incorrect Question 9 0/1 pts The molar solubility of lead () chloride (PbCl2) is 1.6x102 M. What is the Ksp of PbCl2? 4.1x10-6 5.0x10-4 1.6x10-2 3.1x107 1.6x10-5
please show all work. i need to know process for test A compound decomposes by a...
 please show all work. i need to know process for test
A compound decomposes by a first-order process. If 60% of the compound remains after 60 minutes, the half-life of the compound is minutes. -5 O 44 0 -18 45 81
please show all work. i need to know process for test
A compound decomposes by a first-order process. If 60% of the compound remains after 60 minutes, the half-life of the compound is minutes. -5 O 44 0 -18 45 81
Question 4 07 1.5 pts What is the concentration of Pb2+ in a saturated solution of...
 Question 4 07 1.5 pts What is the concentration of Pb2+ in a saturated solution of PbCl2 in 0.10 M KCl solution. (Ksp = 1.4 x 108)? Why is [Pb2') in this question (smaller/larger) than in Question 3? Hint: What is the initial [CI] before PbCl2 is dissolved? because the presence of chloride ion prevents the dissolution 1.4 x 10M of PbCl2 1.5 x 10M same as in Question 3 1.4 x 10-M, because the presence of chloride ion slows...
Question 4 07 1.5 pts What is the concentration of Pb2+ in a saturated solution of PbCl2 in 0.10 M KCl solution. (Ksp = 1.4 x 108)? Why is [Pb2') in this question (smaller/larger) than in Question 3? Hint: What is the initial [CI] before PbCl2 is dissolved? because the presence of chloride ion prevents the dissolution 1.4 x 10M of PbCl2 1.5 x 10M same as in Question 3 1.4 x 10-M, because the presence of chloride ion slows...
Question 22 (4 points) Saved The Ksp of Pbs is 2.5x10-18. What concentration of S2- is...
 Question 22 (4 points) Saved The Ksp of Pbs is 2.5x10-18. What concentration of S2- is necessary to precipitate Pbs from a solution which is 0.030 M in Pb2+? O 8.3x10-17 7.5x10-17 O 8.3x10-19 O 7.5x10-19
Question 22 (4 points) Saved The Ksp of Pbs is 2.5x10-18. What concentration of S2- is necessary to precipitate Pbs from a solution which is 0.030 M in Pb2+? O 8.3x10-17 7.5x10-17 O 8.3x10-19 O 7.5x10-19
Most questions answered within 3 hours.
-
a
dairy farmer notices that a citcular water trough near the barn has
become rusty with...
asked 1 minute ago -
In a study of the accuracy of fast food drive-through orders,
Restaurant A had 283 accurate...
asked 3 minutes ago -
A share of common stock has just paid a dividend of $2.50. If
the expected long-run...
asked 12 minutes ago -
1. List all actors whose last names start
with letter "K"? The output needs to include the actor's...
asked 15 minutes ago -
1.The New England Merchants Bank Building in Boston is 152 m
high. On windy days it...
asked 18 minutes ago -
Can someone explain this code with comments I am supposed to dispay an array an add...
asked 32 minutes ago -
MKT2283 Sales Management Week 2 Assignment
Making Sales Management Decisions – Pronto Retail
Centers
Sales Management,...
asked 39 minutes ago -
Uranium is distributed in a form called yellow cake, which is
made from uranium ore. In...
asked 50 minutes ago -
Python:
Write a func that will create a list of random numbers. The
function will take...
asked 53 minutes ago -
Which of the following statements below is correct? There might
be more than one correct answer....
asked 56 minutes ago -
You find that the annual standard deviation of a stock's returns
is equal to 35%. For...
asked 1 hour ago -
A 2,000 kg car is traveling at 20.0 m/s down a long mountain
grade of 1.00%....
asked 1 hour ago
 please show all work i need to know how to do this for a
test
The reaction AB is first order in (A). Consider the following data. Time (s) 0.0 5.0 10.0 15.0 20.0 [A] (M) 0.20 0.14 0.10 0.071 0.050 The concentration of A is Mafter 40.0 s. 1.3 e-2 O 0.17 O 3.5 e-4 0 0.025 1.2
please show all work i need to know how to do this for a
test
The reaction AB is first order in (A). Consider the following data. Time (s) 0.0 5.0 10.0 15.0 20.0 [A] (M) 0.20 0.14 0.10 0.071 0.050 The concentration of A is Mafter 40.0 s. 1.3 e-2 O 0.17 O 3.5 e-4 0 0.025 1.2
 need help ASAP
3 pts Question 2 Acetaminophen is a medication used to treat pain and fever and contains Carbon, Hydrogen, Oxygen, and Nitrogen. Combustion analysis of a 15.0-mg sample of acetaminophen produced 8.10 mg of water. What is the percent by mass of Hydrogen in acetaminophen? 4.4% 12.0% 7.5% 3.0% 6.0% Question 4 1.5 pts AgCl, FeCO3. Hg Bry, and Pbly are compounds that are insoluble in water. Which compound would be more soluble in acidic solution? AgCl only...
need help ASAP
3 pts Question 2 Acetaminophen is a medication used to treat pain and fever and contains Carbon, Hydrogen, Oxygen, and Nitrogen. Combustion analysis of a 15.0-mg sample of acetaminophen produced 8.10 mg of water. What is the percent by mass of Hydrogen in acetaminophen? 4.4% 12.0% 7.5% 3.0% 6.0% Question 4 1.5 pts AgCl, FeCO3. Hg Bry, and Pbly are compounds that are insoluble in water. Which compound would be more soluble in acidic solution? AgCl only...
 Question 3 7.5 pts Time Running Attempt due: Jur 1 Hour, 14 Mi The concentration of Agt in a solution saturated with Ag2C2O4(s) is 1.50x10^-4 M. Calculate Ksp for AgzC204. Ksp = es
Question 3 7.5 pts Time Running Attempt due: Jur 1 Hour, 14 Mi The concentration of Agt in a solution saturated with Ag2C2O4(s) is 1.50x10^-4 M. Calculate Ksp for AgzC204. Ksp = es
 please show work. i need to know how to do this for a
test
A flask is charged with 0.124 mol of A and allowed to react to form B according to the reaction A(g) →B(g). The following data are obtained for [A] as the reaction proceeds: Time (s) 0.00 10.0- 20.0 30.0 40.0 Moles of A 0.124 0.110 0.088 0.073 0.054 The average rate of disappearance of A between 10 s and 20 s is mol/s. ect Answer 2.2...
please show work. i need to know how to do this for a
test
A flask is charged with 0.124 mol of A and allowed to react to form B according to the reaction A(g) →B(g). The following data are obtained for [A] as the reaction proceeds: Time (s) 0.00 10.0- 20.0 30.0 40.0 Moles of A 0.124 0.110 0.088 0.073 0.054 The average rate of disappearance of A between 10 s and 20 s is mol/s. ect Answer 2.2...
 please show work. i need this for a test
Question 21 1 pts The reaction AB is first order in A. Consider the following data: Time (s) [A] (M) 0.0 2.3 10.0 0.58 20.0 0.14 The rate constant for this reaction is s-1 0.013 O 0.030 O 3.1e-3 O 0.14 O 3.0
please show work. i need this for a test
Question 21 1 pts The reaction AB is first order in A. Consider the following data: Time (s) [A] (M) 0.0 2.3 10.0 0.58 20.0 0.14 The rate constant for this reaction is s-1 0.013 O 0.030 O 3.1e-3 O 0.14 O 3.0
 Please explain the correct
answer for both questions. Thank you!
Incorrect Question 7 0/1 pts The concentration of iodide ions in a saturated solution of lead (11) iodide is_ The solubility product constant of Pblz is 1.4x10-8. 3.5x10-9 3.0x10-3 1.5x103 1.4x10-8 3.8x10-4 Incorrect Question 9 0/1 pts The molar solubility of lead () chloride (PbCl2) is 1.6x102 M. What is the Ksp of PbCl2? 4.1x10-6 5.0x10-4 1.6x10-2 3.1x107 1.6x10-5
Please explain the correct
answer for both questions. Thank you!
Incorrect Question 7 0/1 pts The concentration of iodide ions in a saturated solution of lead (11) iodide is_ The solubility product constant of Pblz is 1.4x10-8. 3.5x10-9 3.0x10-3 1.5x103 1.4x10-8 3.8x10-4 Incorrect Question 9 0/1 pts The molar solubility of lead () chloride (PbCl2) is 1.6x102 M. What is the Ksp of PbCl2? 4.1x10-6 5.0x10-4 1.6x10-2 3.1x107 1.6x10-5
 please show all work. i need to know process for test
A compound decomposes by a first-order process. If 60% of the compound remains after 60 minutes, the half-life of the compound is minutes. -5 O 44 0 -18 45 81
please show all work. i need to know process for test
A compound decomposes by a first-order process. If 60% of the compound remains after 60 minutes, the half-life of the compound is minutes. -5 O 44 0 -18 45 81
 Question 4 07 1.5 pts What is the concentration of Pb2+ in a saturated solution of PbCl2 in 0.10 M KCl solution. (Ksp = 1.4 x 108)? Why is [Pb2') in this question (smaller/larger) than in Question 3? Hint: What is the initial [CI] before PbCl2 is dissolved? because the presence of chloride ion prevents the dissolution 1.4 x 10M of PbCl2 1.5 x 10M same as in Question 3 1.4 x 10-M, because the presence of chloride ion slows...
Question 4 07 1.5 pts What is the concentration of Pb2+ in a saturated solution of PbCl2 in 0.10 M KCl solution. (Ksp = 1.4 x 108)? Why is [Pb2') in this question (smaller/larger) than in Question 3? Hint: What is the initial [CI] before PbCl2 is dissolved? because the presence of chloride ion prevents the dissolution 1.4 x 10M of PbCl2 1.5 x 10M same as in Question 3 1.4 x 10-M, because the presence of chloride ion slows...
 Question 22 (4 points) Saved The Ksp of Pbs is 2.5x10-18. What concentration of S2- is necessary to precipitate Pbs from a solution which is 0.030 M in Pb2+? O 8.3x10-17 7.5x10-17 O 8.3x10-19 O 7.5x10-19
Question 22 (4 points) Saved The Ksp of Pbs is 2.5x10-18. What concentration of S2- is necessary to precipitate Pbs from a solution which is 0.030 M in Pb2+? O 8.3x10-17 7.5x10-17 O 8.3x10-19 O 7.5x10-19